Abstract
Chronic inflammatory diseases are disorders multifactorial growing on elderly and diabetic populations. 15-deoxy-delta12,14-prostaglandin J2 (15d-PGJ2) acts in protective role against oxidative stress and inflammation both in vivo and in vitro.
Here, we present a simple method to incorporate 15d-PGJ2 nanoparticles in acellular human amniotic membrane scaffold, a potential local anti-inflammatory delivery system. After completely removing the cells on the amniotic membrane with a sodium dodecyl sulphate and mechanical approach, we seeded Vero cells incorporated with 15d-PGJ2 nanoparticles. The morphology of the Vero cells and nanoparticles was observed by scanning electron microscopy (SEM) and phase contrast microscopy. The cells cultivated presented the incorporation of the nanoparticles, smooth surface and spherical shape.
Within this study it was shown that amniotic membrane can be used to incorporate cells and anti-inflammatory loaded nanoparticles for prevention and treatment of various diseases.
Introduction
The incidence of people with chronic inflammatory diseases has been increasing over the last three decades, threatening human health. A number of factors are recognized as causes of the pathogenesis as for example the autoimmune diseases, metabolic disorders and chronic respiratory disease [1].
The 15-Deoxy-Δ12,14-Prostaglandin J2 (15d-PGJ2) is a natural ligand of peroxisome proliferator activated receptor γ(PPARγ). This molecule has anti-inflammatory property and is involved in a variety of physiological and pathological processes, including rheumatoid arthritis, myocardial infarction, neural damage, and tumorigenesis [2].
The nanomaterials could provide a revolution in technology that will soon impact the diseases treatment methods through new nanoparticles delivery systems [3]. The development of new strategies for regenerative medicine is one of the most active research in the areas of nanotechnology[4].
Current advances in biotechnology uses acellular scaffolds to regenerate tissues, this is an approach highly beneficial in terms of biocompatibility and biofunctionality [5]. The decellularized human amniotic membrane is the commonest sources for scaffolds used in tissue regeneration because of it good biocompatibility and biodegradability [6].
In this study, we aimed to incorporate 15-deoxy-Δ12,14-PG J2 nanoparticles in acellular human amniotic membrane scaffold as a potential local anti-inflammatory delivery system.
Methods
Preparation of acellu2021 Copyright OAT. All rights reserv
The study was approved by the Hospital Pequeno Príncipe Ethical Committee for the usage of biological material for research purposes approved under article number 0948-11. All materials were used in compliance with ethical guidelines by the Brazilian National Health Council. Fresh human amniotic membrane (HAM) was obtained after caesarian deliveries. Maternal donors provided informed consent and were serologically negative for HIV, hepatitis B, hepatitis C, and syphilis. Briefly, blood clots were immediately cleaned off the placenta after surgery with phosphate buffered saline (PBS) solution containing 100 U/mL penicillin and 100 mg/mL streptomycin.
The acellular human amniotic scaffold (AHAS) was prepared as described by Riau et al., 2010 [7]. Part of the human HAM was then deprived of amniotic epithelial cells to obtain AHAS by sodium dodecyl sulphate in PBS, and incubated with shaking rate of 100 rpm at 37°C for 24 h. Finally, the prepared AHAS was rewashed 3 times with PBS.
Nanoencapsulation of 15d-PGJ2 in poly(D,L-lactide-co-glycolide) (PLGA) nanocapsules (15d-PGJ2-NC). The 15d-PGJ2-NC were prepared by the nanoprecipitation method, as described by Fessi et al., 1989, and supplied by Dr. Napimoga from Laboratory of Immunology and Molecular Biology, São Leopoldo Mandic Institute and Research Center [8,9].
Culture and seeding of VERO cells with 15d-PGJ2-NC
Vero cells (ATCC® CCL-81™) were cultured for 7 days in DMEM (Dulbecco’s modified Eagle medium, Invitrogen) supplemented with 10% FBS (fetal bovine serum, Invitrogen) and 100 U/mL penicillin and 100 mg/mL streptomycin at 37°C with 5% CO2. The culture medium was changed every 2 days. The Vero cells were seeded on plastic plate wells over AHAS and with 15d-PGJ2-NC in concentration of 10 µg/mL for 24h in order to cells incorporate the nanoparticles, after the incubation time the plate was washed with PBS. Phase contrast images were obtained with Axio Vert.A1 (Zeiss).
Scanning electron microscopy (SEM)
The morphology and structure of the cells with 15d-PGJ2-NC cultivated in AHAS were examined in a JEOL 1200EX II microscope (Jeol ltda, Akishima) operating at 80 kV. In order to perform the SEM analysis, material was fixed on top coverslip, dried, mounted on a stub for SEM, fixed in 2.5% (v/v) glutaraldehyde (Sigma-Aldrich) in PBS and post-fixed with 1% (v/v) and 0.1 M sodium cacodylate trihydrate (Sigma-Aldrich).
Results
SEM morphology
Nanoparticles characteristics of 15d-PGJ2 in the Vero cells cultivated on acellular amniotic membrane were revealed by SEM analysis. Figure 1 shows the vero cells cultivated on AHAS (a), Vero cells incubated with 15d-PGJ2 10 µg/mL (b). Vero cell cultivated on AHAS and incubated with 15d-PGJ2 10 µg/mL at 6.500x (c) and 13.000x (d). The presence of 15d-PGJ2 in the surface the amniotic membrane can be noticed. At 15d-PGJ2 concentration (10 µg/mL), a more uniform covering of the cell is observed.
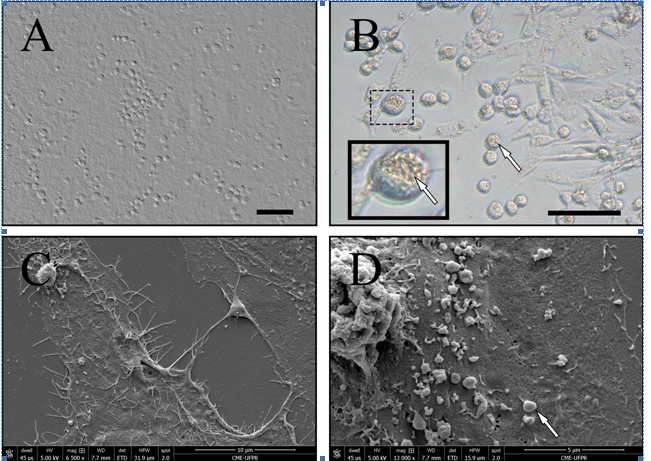
Figure 1. Phase contrast images of Vero cells in amniotic membrane after 24 hours. (A): Vero cells plated on amniotic membrane. (B): Morphological rearrangement of cell 24h post- incubated with 15d-PGJ2 nanoparticles with the formation of homogeneous spherical aggregates (note the magnified image of particle agglomerate [arrow]). (c): The SEM images show Vero cells on acellular amniotic membranes cultured after 24 h: Cells on amniotic membrane and nanoparticles with cells on membranes (d,) (note the cell with nanoparticles (15d-PGJ2) on cell membrane [arrow]). The bar scale is 50 μm.
Discussion
In recent decades, the nanotechnology has progressed rapidly. One of the most important factors in regenerative medicine is the preparation of a scaffold. Acellular amniotic membrane human scaffolds have recently become the focus of interest mainly due to the possible beneficial and applications in regenerative medicine [10].
So far, various decellularization methods have been suggested to develop extracellular matrix derived scaffolds. Tissue engineering using acellular scaffods has introduced a new field of repair in the treatment of wounds tissues or diseases [11].
HAM is an appropriate substitute for reconstruction of various organs and tissues due to its availability, low cost and low risk of viral disease transmission and immunologic rejection [12]. The overall aim of this study was to develop a type of surface covered by nanoparticles and cells with tissue engineering applications in the form of a 3D scaffold.
The use of chemical approach for decellularization shown in SEM completely removal of cells while leaving the matrix intact, unlike in other studies[13].
An intact extracellular matrix is essential for recellularization, because the extra cellular matrix contains components necessary for cell attachment, proliferation, and ultimately tissue remodeling [14]. In this study, we demonstrated the recellularization efficiency with the cultivation of VERO cells in AHAS provide an adequate in vitro microenvironment for proliferation and incorporation 15d-PGJ2 nanoparticles in acellular scaffold.
Conflict of interest
None declared.
Conclusion
15-deoxy-Δ12,14-prostaglandin J2 (15d-PGJ2) is one of the potent ligand of peroxisome proliferator-activated receptor γ (PPARγ), suppresses proliferation and induces intracellular apoptosis in different events in excessive inflammation. This study showed that AHAS could be used as a biomaterial and be applied as a scaffold for regenerative medicine and nanoparticle delivery.
References
- Tuttolomondo A, Maida C, Pinto A (2015) Diabetic foot syndrome: Immune-inflammatory features as possible cardiovascular markers in diabetes. World Journal of Orthopedic 6: 62-76.
- Buckner MM, Antunes LC, Gill N, Russell SL, Shames SR, et al. (2013) 15-Deoxy-Delta12,14-prostaglandin J2 inhibits macrophage colonization by Salmonella enterica serovar Typhimurium. PloS One 8: e69759. [Crossref]
- Clemente-Napimoga JT, Moreira JA, Grillo R, de Melo NF, Fraceto LF, et al. (2012)15d-PGJ2-loaded in nanocapsules enhance the antinociceptive properties into rat temporomandibular hypernociception. Life Sciences 90: 944-949.
- Huynh NT, Passirani C, Saulnier P, Benoit JP (2009) Lipid nanocapsules: a new platform for nanomedicine. Int J Pharm 379: 201-209. [Crossref]
- Garg T, Singh O, Arora S, Murthy R: Scaffold (2010) A novel carrier for cell and drug delivery. Crit Rev Ther Drug Carrier Syst 29: 1-63. [Crossref]
- Huang G, Ji S, Luo P, Liu H, Zhu S, et al. (2013) Accelerated expansion of epidermal keratinocyte and improved dermal reconstruction achieved by engineered amniotic membrane. Cell Transplant 22: 1831-1844. [Crossref]
- Riau AK, Beuerman RW, Lim LS, Mehta JS (2010) Preservation, sterilization and de-epithelialization of human amniotic membrane for use in ocular surface reconstruction. Biomaterials 31: 216-225. [Crossref]
- Fessi H PF, Devissaguet JP, Ammoury N, Benita S (1989) Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int J Pharm 55: R1-R4.
- Alves C, de Melo N, Fraceto L, de Araujo D, Napimoga M: Effects of 15d-PGJ(2)-loaded poly(D,L-lactide-co-glycolide) nanocapsules on inflammation. Br J Pharmacol 162: 623-632. [Crossref]
- Diaz-Prado S, Rendal-Vazquez ME, Muinos-Lopez E, Hermida-Gomez T, Rodriguez-Cabarcos M, et al. (2010) Potential use of the human amniotic membrane as a scaffold in human articular cartilage repair. Cell and Tissue Banking 11: 183-195.
- Debels H, Hamdi M, Abberton K, Morrison W (2015) Dermal matrices and bioengineered skin substitutes: a critical review of current options. Plast Reconstr Surg Glob Open 3: e284. [Crossref]
- Niknejad H, Peirovi H, Jorjani M, Ahmadiani A, Ghanavi J, et al. (2008) Properties of the amniotic membrane for potential use in tissue engineering. Eur Cell Mater 15: 88-99. [Crossref]
- Wilshaw SP, Kearney JN, Fisher J, Ingham E (2006) Production of an acellular amniotic membrane matrix for use in tissue engineering. Tissue Eng12: 2117-2129. [Crossref]
- Luo JC, Li XQ, Yang ZM (2004) Preparation of human acellular amniotic membrane and its cytocompatibility and biocompatibility. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 18: 108-111. [Crossref]

