Abstract
Neutrophilic dermatoses (NDs) are a heterogeneous group of disorders with neutrophil infiltration, variable degrees of leukocytoclasia and the absence of vasculitis as common pathologic pictures. These disorders often are idiopathic, but their association with hematological and solid malignancies, auto-inflammatory diseases, autoimmune connective tissue diseases, infections, medications, and vaccinations has been reported.
Herein, we report a case of ND in association with ankylosing spondylitis and review the literature regarding the association of this dermatosis with inflammatory autoimmune disorders.
Introduction
Neutrophilic dermatoses [NDs] which were initially described in 1991 [1], are a heterogeneous group of disorders with dermal neutrophil infiltration [1,2], variable degrees of leukocytoclasia and the absence of vasculitis [3] as common histologic features. Although most cases of ND are idiopathic [1,2,4,5], the association with hematological and solid malignancies, auto-inflammatory diseases, autoimmune connective tissue diseases [1,2,4-6], infections, medications [1,2,4,5,7], and vaccinations [1,4] are well known. Among medications, granulocyte colony-stimulating factor [GCSF] [1,2], tumor necrosis factor [TNF] -targeting agents [4,8], all-trans-retinoic acid proteosome inhibitors, hypomethylating agents, tyrosine kinase inhibitors, lenalidomide, vemurafenibm, ipilimumab, cyclines, trimethoprim-sulfamethoxazole, oral contraceptives, furosemide, celecoxib, azathioprine and propylthiouracil are the most important therapeutic agents that have been associated with ND [1]. In ND cases, low hemoglobin levels [1], and involvement of internal organs [1,2] are markers for high suspicious of associated malignancies.
Currently, INF-α inhibitors such as etanercept play a great role in treating autoimmune and connective tissue diseases [4]. A spectrum of adverse effects have been reported with these agents including induction or triggering of autoimmune diseases [4,9,10], neutrophilic dermatoses [4,8,11-13], psoraisiform eruptions, and granulomatous conditions [10].
Ankylosing spondylitis [AS] is an inflammatory spondiloarthropathy which mainly involves the spine and sacroiliac joints resulting in new bone formation, syndesmophytes, and ankylosis of the vertebral column [14]. To our best knowledge, ND in association with AS has been reported in few cases. Herein, we report a case of this association and review literature regarding the association of ND with inflammatory autoimmune disorders.
Case report
A 33-year old man was referred to us with a pruritic skin eruption since 3 months ago (Figure 1). The patient has carried a diagnosis of ankylosing spondylitis (AS) for 15 years. He was under treatment with etanercept and prednisolone. His lesions started in his groin, then gradually spread to his trunk and extremities. The pruritus severity was more at night. He complained of fever and aggravation of articular pains during these 3 months.
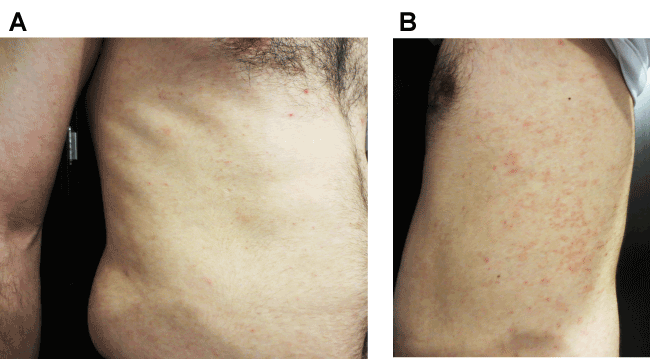
Figure 1. Many tiny targetoid erythematous papules aggregated in extremities and trunk
During this period, before coming to our attention, therapeutic agents against scabies had been prescribed for him for many times, with no demonstrable benefit. With a probable diagnosis of drug reaction, etanercept therapy had been stopped by a dermatologist, but again the lesions remained.
On physical examination, there were many tiny, tender, targetoid erythematous papules aggregated in the groin, genitalia, extremities and trunk. A biopsy was taken from the lesions with differential diagnoses of erythema multiforme, hypersensitivity or drug reactions, and leukocytoclastic vasculitis.
Histologic sections demonstrated skin with an unremarkable appearing epidermis (Figure 2). Within the reticular dermis, there was a dense and diffuse infiltrate of neutrophils. Rare eosinophils were also present. The infiltrate was not perivascular and didn’t not invade nor destroy vessel walls. Extravasated erythrocytes were not present and there was no fibrin in the vessel walls. Necrosis was not seen. The histologic features were those of a neutrophilic dermatosis such as that described in Sweet’s syndrome (SS) and as occurring in conjunction with connective tissue disorders such as rheumatoid arthritis (RA) and systemic lupus erythematous (SLE).
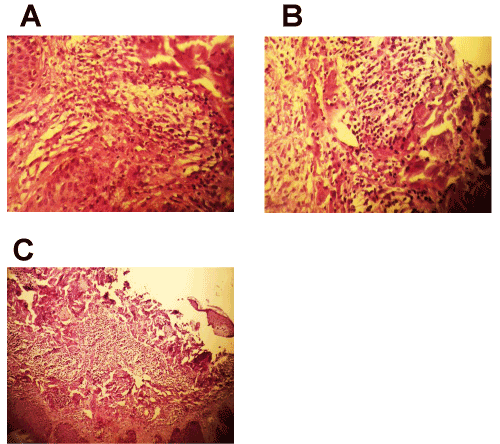
Figure 2. In pathological aspect, there is a dense and diffuse infiltrate of neutrophils and rare eosinophils within the reticular dermis
According to the pathological report and clinical manifestation, the diagnosis of ND was suggested. We considered if this dermatosis was associated with etanercept-related adverse reaction or was secondary to the underlying steoarthropathy. Laboratory factors of disease activity were checked. Upon blood analysis, a neutrophilia was seen. Elevated levels of C-reactive protein, rheumatoid factor, and erythrocyte sedimentation rate were seen. Because of the presence of positive clues for AS activation and continuation of lesions despite etanercept withdrawal, the diagnosis of Sweet’s Syndrome secondary to AS was the most probable diagnosis. Hence, for the patient, prednisolone (20 mg/day) and topical clobetasol were prescribe. Lesions showed recovery about 10 days after starting the treatment.
Discussion
The NDs are a heterogeneous group of disorders with neutrophil infiltration as common pathologic picture [1,2]. A variety of types of these dermatoses have been reported. In the following, their most important types and their association with inflammatory and connective tissue diseases have been summarized:
Sweet's syndrome (SS) [1,2]: SS which is known as acute febrile neutrophilic dermatosis, is characterized by tender erythematous papules, plaques, and nodules, fever, malaise, arthralgia, and neutrophilia [1,2,5,7,15]. On histologic sections, a diffuse and band like infiltrations of neutrophils [1] localized in the upper dermis is seen, which can extend to the subcutaneous tissue [2]. Systemic lupus erythematosus [1,2], rheumatoid arthritis [1,2,5,10,16], Behçet disease [2], Sjögren's syndrome [5], relapsing polychondritis, erythema nodosum, Crohn's disease, Hashimoto and Grave's thyroiditis [1, 2] are among the disorders with well-known associations with SS. We found only four cases of Sweet’s syndrome associated with AS in searching the computerized bibliographic database PubMed [6,17-19]. It seems our case is the fifth case which shows association of AS with this type of neutrophilic dermatosis.
Pyoderma gangrenosum (PG) [1, 2]: PG is characterized by ulcerative lesions with typical violaceous undermined borders [1,2,7]. In most cases, its pathological manifestations are not specific and suggest the diagnosis of infection [1]. Its association with SLE [1,2], RA [1,2,10], juvenile RA [20], AS [2], relapsing polychondritis, erythema nodosum, Crohn's disease [1, 2], Hashimoto and Grave's thyroiditis [1] has been reported.
Rheumatoid neutrophilic dermatitis (RND) [2, 15, 21, 22]: RND, which was introduced by Ackerman [15, 21], is seen in patients with severe, seropositive RA [2,10,15,21,22]. It presents as urticarial-like papules, plaques, and nodule, [15] which typically involve the skin overlying the joints in a symmetric manner [3, 15]. Histologically, there is a dense, diffuse neutrophilic infiltration involving the whole dermis and often extending into the subcutaneous fat [15,21].
Neutrophilic eccrine hidradenitis (NEH] [1,2]: NEH is another type of ND with peri-eccrine gland neutrophilic infiltration, which involves the face, upper extremities, palms and soles [1]. This disorder is seen in patients receiving chemotherapeutic agents for acute myelogenous leukemia [2].
Neutrophilic panniculitis [NP] [2,23]: NP is characterized by tender subcutaneous nodules and abscesses [2]. An association with RA [2,24], juvenile RA [23], myelodysplastic syndrome [2,23], and Crohn’s disease [2] has been reported.
Subcorneal pustular dermatosis (SPD) or Sneddon-Wilkinson disease [1,2]: SPD is characterized by flaccid pustules grouped in annular configurations involving the trunk and flexural areas [1]. Histologically, neutrophil accumulation in the upper epidermis is the predominant feature [7]. Its association with RA, SLE, Sjögren syndrome, SAPHO [synovitis, acne, pustulosis, hyperostosis, osteitis] syndrome, multiple myelomas, monoclonal gammopathies, and lymphomas has been reported [2].
Erythema elevatum diutinum [EED] [1,2]: EED presents as tender purplish papules or nodules on the extensor aspects of extremities [1,2,15]. Microscopically, leukocytoclastic vasculitis is the predominant presentation in the early stages [1]. Its association with connective tissue diseases particularly RA has been reported [2].
Palisaded neutrophilic granulomatous dermatitis [PNGD] [4]: PNGD is a rare type of ND [4], which was introduced by Dykman et al in 1965 [25]. Today, some concepts such as interstitial granulomatous dermatitis with arthritis, linear subcutaneous bands, rheumatoid papules, rheumatoid granuloma, linear granuloma annulare, superficial ulcerating rheumatoid necrobiosis, necrobiotic granuloma, palisading granuloma, cutaneous extravascular necrotizing granuloma, and Churg-Strauss granuloma are included in this classification [25,26]. Clinically this dermatosis is characterized by a variety of presentations ranging from asymptomatic papules, plaques and nodules to indurated linear bands, urticarial lesions, petechiae reticularis, and livedo reticularis. Tenderness and ulceration has been reported in patients [4]. Its pathologic manifestations vary from a sparse perivascular infiltration of neutrophils, lymphocytes, histiocytes, and eosinophils, to interstitial or palisaded granuloma formation along with dermal fibrosis [4,25]. Upon immunofluorescence studies, deposition of IgM and C3 are seen in small vessels [26].
The association of PNGD with connective tissue disease, autoimmune inflammatory diseases, and infections has been reported. RA [4,12,13,22,25-29], SLE [4,25-29], systemic sclerosis [4,29], Behçet's disease [27,29,30], systemic vasculitis [4,25-28], inflammatory bowel disease [4,26,28], sarcoidosis [4,27], lymphoproliferative disorders [4,25-28], Wegener's granulomatosis [26], and bacterial endocarditis [26,27] are the most important associations. There are some reports concerning the association of PNGD with AS [4]. The appearance of PNGD lesions seems to be related with worsening of the underlying systemic disease [25]. In addition, the association of this dermatitis with TNF-α inhibitors such as etanercept, infliximab, and adalimumab has been reported [4,11-13].
Non-bullous neutrophilic dermatosis (NND) [3]: NBND which also known as non-bullous neutrophilic LE, and Sweet-like ND is a recently-introduced ND and is characterized as urticarial lesions in context of connective tissue disorders. Microscopically, interstitial and perivascular neutrophilic infiltration, leukocytoclasia, and vacuolar alteration along the dermal-epidermal junction are the predominant presentations. It has been reported in association with SLE, RA, and Sjögren syndrome [3].
Although the association of ND with autoimmune connective tissue and inflammatory diseases has been reported in literature, the question that this association is a real concept or only an accident coincidence hasn’t been answered. Additionally, the point that patients with autoimmune connective tissue disorders receive many biologic therapeutic agents that may induce a ND, complicates the answer to this question. If we propose this association as a real one, the exact pathogenic association is not yet been understood, because the exact pathogenesis of these diseases hasn’t been clarified [2,19]. Some hypotheses have been suggested:
- Deposition of immunoglobulins, complement, and fibrinogen in the areas of altered collagen and vessel wall in the context of connective tissue diseases results in immune complex-mediated vasculitis [19,25] through a mechanism similar to the type III hypersensitivity reactions [19]. This immune complex deposition can trigger the appearance of NDs through neutrophil recruitment [21,25,26].
- Impaired function of neutrophils has been reported as a common pathway in pathogenesis of connective tissue disorders and NDs [19]. Studies have shown the role of neutrophilic recruitment in inducing chronic inflammation in the context of autoimmune connective tissue disorders [2,31]. Hence, NDs with neutrophilic activation can occur in association with these disorders [2].
- Dysregulated profile of cytokines plays a role in association of connective tissue disorders and NDs [19]. The expression of interleukin [IL]-1ß and its receptor, IL-8, IL-17, TNF-α, CXCL1/2/3, CXCL16, and metalloproteinases 2 and 9 are up-regulated in skin lesions of ND [1], among them the roles of IL- 1 [1,2] and TNF-α [32] are significant. In the serum of these patients, elevated levels of IL-1, interferon [INF]-γ and GCSF have been reported [1]. In connective tissue diseases, IL-1ß [33], IL-1 receptor 2 [34], TNF [33], and IL-23/IL-17 pathway [35] play roles in the pathogenesis. This dysregulated cytokine profile results in neutrophil recruitment in both groups of these disorders [1].
- T-cell-dependent cellular immune activation during the active phase of connective tissue diseases has been suggested as a cause for triggering NDs [19]. It appears T cells play a role in recruiting mature neutrophils to the dermis [1,32]. It has been reported that cytokines such as IL-17, which have a role in T cell polarization, are overexpressed in skin lesions of cases with ND and connective tissue diseases [2].
- Infectious agents have been suggested as a common pathogenic factor for connective tissue diseases and NDs [19].
- In context of NDs, articular involvement has been reported, which can result in arthralgia. Although this articular involvement is different from the one in the context of connective tissue diseases in terms of clinical presentation and radiologic pattern [2], sometimes they cannot be differentiated. In this situation, articular involvements secondary to ND are misdiagnosed as ND in association with the connective tissue diseases.
The list of differential diagnoses of NDs includes urticaria [4], neutrophilic urticaria [15], leukocytoclastic vasculitis [4], urticarial vasculitis [15], interstitial granuloma annulare [4], dermatitis herpetiformis, cellulitis [21], erythema multiforme, hypersensitivity and drug reactions.
In some cases, ND is self-limiting [4,25]. Some therapeutic agents and modalities have successfully been used (Table 1) [1,2,4,7,21,25,32,36-38]. Although the role of TNF-α inhibitors in triggering NDs has been well known, they are first choice for treating ND in association with inflammatory bowel disease or RA [1].
Table 1. List of therapeutic agents and modalities which have successfully been used in treating NDs
Topical and oral corticosteroids [1,2,4,7,21,25, 32]
Dapsone [1,4,7,21,25]
Cyclosporine [1,4,7],
Hydroxychloroquine [4,21,25,36]
Colchicine [1,7,32]
Potassium iodide [1,7,32]
Indomethacin [1,7,32]
Clofazimine [1,7,32]
TNF-α inhibitors [1,7,32,37]
IL-1 receptor antagonists (anakinra) [1,2,38]
IL-6 blocking agents (tocilizumab) [2]
Thalidomide [1,32]
Cytotoxic chemotherapies and antimetabolites such as methotrexate,azathioprine,cyclophosphamide and mycophenolate mofetil [1,32]
Systemic retinoids [1,7]
Intravenous immunoglobulin[1,2]
Topical calcineutin inhibitors [1]
Phototherapy [7]
Hyperbaric oxygen [7]
Plasmapheresis [7] |
Conclusion
As cases with connective diseases receive a verity of medications, the question as to the whether the association between these disorders and NDs is a real one or if the NDs appear as side effects of medications still hasn’t been fully answered.
References
- Maalouf D, Battistella M, Bouaziz JD (2015) Neutrophilic dermatosis: disease mechanism and treatment. Curr Opin Hematol 22: 23-29. [Crossref]
- Prat L, Bouaziz JD, Wallach D, Vignon-Pennamen MD, Bagot M1 (2014) Neutrophilic dermatoses as systemic diseases. Clin Dermatol 32: 376-388. [Crossref]
- Saeb-Lima M, Charli-Joseph Y, RodrÃguez-Acosta ED, DomÃnguez-Cherit J (2013) Autoimmunity-related neutrophilic dermatosis: a newly described entity that is not exclusive of systemic lupus erythematosus. Am J Dermatopathol 35: 655-660. [Crossref]
- de Unamuno Bustos B, Rabasco AG, Sánchez RB, de Miquel VA (2013) Palisaded neutrophilic and granulomatous dermatitis associated with ankylosing spondylitis. Am J Dermatopathol 35: 847-850. [Crossref]
- Mrabet D, Saadi F, Zaraa I, Chelly I, Sahli H, et al. (2011) Sweet's syndrome in a patient with rheumatoid arthritis, Sjögren's syndrome and lymph node tuberculosis. BMJ Case Rep 2: 2011. [Crossref]
- Petermann A, Tebbe B, Distler A, Sieper J, Braun J (1999) Sweet's syndrome in a patient with acute Crohn's colitis and longstanding ankylosing spondylitis. Clin Exp Rheumatol 17: 607-610. [Crossref]
- Cohen PR1 (2009) Neutrophilic dermatoses: a review of current treatment options. Am J Clin Dermatol 10: 301-312. [Crossref]
- Roe E, Puig L, Corella F, GarcÃa-Navarro X, Alomar A (2008) Cutaneous adverse effects of biological therapies for psoriasis. Eur J Dermatol 18: 693-699. [Crossref]
- Ramos-Casals M, Brito-Zerón P, Muñoz S, Soria N, Galiana D, et al. (2007) Autoimmune diseases induced by TNF-targeted therapies: analysis of 233 cases. Medicine (Baltimore) 86: 242-251. [Crossref]
- Xue Y, Cohen JM, Wright NA, Merola JF (2016) Skin Signs of Rheumatoid Arthritis and its Therapy-Induced Cutaneous Side Effects. Am J Clin Dermatol 17: 147-162. [Crossref]
- Deng A, Harvey V, Sina B, Strobel D, Badros A, et al. (2006) Interstitial granulomatous dermatitis associated with the use of tumor necrosis factor alpha inhibitors. Arch Dermatol 142: 198-202. [Crossref]
2021 Copyright OAT. All rights reserv
- Collaris EJ, van Marion AM, Frank J, Poblete-Gutiérrez P (2007) Cutaneous granulomas in rheumatoid arthritis. Int J Dermatol 46 Suppl 3: 33-35. [Crossref]
- Stephenson SR, Campbell SM, Drew GS, Magro CM (2011) Palisaded neutrophilic and granulomatous dermatitis presenting in a patient with rheumatoid arthritis on adalimumab. J Cutan Pathol 38: 644-648. [Crossref]
- Kim H, Lee J, Ahn JK, Hwang J, Park EJ, et al. (2015) Predictive factors of radiographic progression in ankylosing spondylitis. Korean J Intern Med 30: 391-397. [Crossref]
- Scherbenske JM, Benson PM, Lupton GP, Samlaska CP (1989) Rheumatoid neutrophilic dermatitis. Arch Dermatol 125: 1105-1108. [Crossref]
- Harary AM (1983) Sweet's syndrome associated with rheumatoid arthritis. Arch Intern Med 143: 1993-1995. [Crossref]
- Wendling D, Léaustic M, Toussirot E, Prati C (2008) Ankylosing spondylitis and Sweet's syndrome. Clin Rheumatol 27 Suppl 1: S27-28. [Crossref]
- El Mahou S, Vandici S, Morel G, Lebas D, Deprez X (2009) Diagnosis of a spondylarthropathy in Sweet,s syndrome. Joint Bone Spine 76: 922-931.
- Mansouri S, Abourazzak FE, Aradoini N, Bettioui A, Fourtassi M, et al. (2013) Ankylosing spondylitis associated with Sweet's syndrome: a case report. J Med Case Rep 7: 16. [Crossref]
- Xue Y, Cohen JM, Wright NA, Merola JF (2016) Skin Signs of Rheumatoid Arthritis and its Therapy-Induced Cutaneous Side Effects. Am J Clin Dermatol 17: 147-162. [Crossref]
- Mashek HA, Pham CT, Helm TN, Klaus M (1997) Rheumatoid neutrophilic dermatitis. Arch Dermatol 133: 757-760. [Crossref]
- Ergun T, Inanc N, Tuney D, Kotiloglu EK, Seckin D, et al. (2008) Skin manifestations of rheumatoid arthritis: a study of 215 Turkish patients. Int J Dermatol 47: 894-902. [Crossref]
- Dyer JA, Guitart J, Klein-Gitelman M, Mancini AJ (2007) Neutrophilic panniculitis in infancy: a cutaneous manifestation of juvenile rheumatoid arthritis. J Am Acad Dermatol 57: S65-S68. [Crossref]
- Tran TA, DuPree M, Carlson JA (1999) Neutrophilic lobular (pustular) panniculitis associated with rheumatoid arthritis: a case report and review of the literature. Am J Dermatopathol 21: 247-252. [Crossref]
- Bremner R, Simpson E, White CR, Morrison L, Deodhar A (2004) Palisaded neutrophilic and granulomatous dermatitis: an unusual cutaneous manifestation of immune-mediated disorders. Semin Arthritis Rheum 34: 610-616. [Crossref]
- Chu P, Connolly MK, LeBoit PE (1994) The histopathologic spectrum of palisaded neutrophilic and granulomatous dermatitis in patients with collagen vascular disease. Arch Dermatol 130: 1278-1283. [Crossref]
- Paštar Z, Radoš J, Pavić I, Kovačević S, Vukasović A, et al. (2013) Palisaded neutrophilic and granulomatous dermatitis in association with subcutaneous nodular and systemic sarcoidosis. Acta Dermatovenerol Croat 21: 245-249. [Crossref]
- Gulati A, Paige D, Yaqoob M, Proby CM, Cerio R, et al. (2009) Palisaded neutrophilic granulomatous dermatitis associated with systemic lupus erythematosus presenting with the burning rope sign. J Am Acad Dermatol 61: 711-714. [Crossref]
- Hantash BM, Chiang D, Kohler S, Fiorentino D (2008) Palisaded neutrophilic and granulomatous dermatitis associated with limited systemic sclerosis. J Am Acad Dermatol 58: 661-664. [Crossref]
- Kim SK, Park CK, Park YW, Jun JB, Yoo DH, et al. (2005) Palisaded neutrophilic granulomatous dermatitis presenting as an unusual skin manifestation in a patient with Behçet's disease. Scand J Rheumatol 34: 324-327. [Crossref]
- Dominical VM, Bértolo MB, Almeida CB, Garrido VT, Miguel LI, et al. (2011) Neutrophils of rheumatoid arthritis patients on anti-TNF-α therapy and in disease remission present reduced adhesive functions in association with decreased circulating neutrophil-attractant chemokine levels. Scand J Immunol 73: 309-318. [Crossref]
- Reguiaï Z, Grange F (2007) The role of anti-tumor necrosis factor-alpha therapy in Pyoderma gangrenosum associated with inflammatory bowel disease. Am J Clin Dermatol 8: 67-77. [Crossref]
- Aradi B, Kato M, Filkova M, Karouzakis E, Klein K, et al. (2015) Protein tyrosine phosphatase nonreceptor type 2: an important regulator of lnterleukin-6 production in rheumatoid arthritis synovial fibroblasts. Arthritis Rheumatol 67: 2624-2633. [Crossref]
- Momenzadeh P, Mahmoudi M, Beigy M, Garshasbi M, Vodjdanian M, et al. (2016) Determination of IL1 R, ANTXR, CARD9, and SNAPC4 single nucleotide polymorphisms in Iranian patients with ankylosing spondylitis. Rheumatol Int 36: 429-435. [Crossref]
- Paine A, Ritchlin CT (2016) Targeting the interleukin-23/17 axis in axial spondyloarthritis. Curr Opin Rheumatol 28: 359-367. [Crossref]
- Gerbing EK, Metze D, Luger TA, Ständer S (2003) Interstitial granulomatous dermatitis without arthritis: successful therapy with hydroxychloroquine. J Dtsch Dermatol Ges 1: 137-141. [Crossref]
- Mahe E, Descamps V (2002) [Anti-TNF alpha in dermatology]. Ann Dermatol Venereol 129: 1374-1379. [Crossref]
- Lipsker D, Lenormand C (2012) Indications and modes of use for interleukin (IL)-1 antagonists in inflammatory dermatosis: a new therapeutic approach to immune-mediated inflammatory diseases. Ann Dermatol Venereol 139: 459-467. [Crossref]


