Cardiovascular disease remains the leading cause of death in men and women both in the US and worldwide. Due to increased life expectancies in developed countries, it is predicted that more individuals will develop cancer, and a significant proportion will have underlying cardiac disease. Similarly, improved survival rates in cancer patients have led to an increased awareness of the presence of and potential worsening of cardiovascular disease in cancer patients. Unfortunately cardiovascular complications can be a common occurrence due to worsening of underlying disease or side effects from cancer therapy [1,2]. Although recent advances have led to improved survival and quality of life from chemotherapy, the increase in life expectancy from cancer agents may be counter balanced by the increased morbidity and mortality from cardiac pathology. These complications can arise due to local invasion of disease or distant metastatic spread, causing syndromes such as superior vena cava syndrome or tamponade. Indirect complications of cancer such as hyperviscosity syndromes can also be seen. Of greater concern, and also of the subject of discussion in this review, is the fact that many of the chemotherapeutic or radiation therapies can be directly toxic to the cardiovascular system. For the optimal care of the cancer patient, an integrated approach by the oncologist, cardiologist, and the primary care provider is essential.
Cardiotoxicity is defined by the National Cancer Institute as “toxicity that affects the heart”. Clarification of this definition has been provided by the Cardiac Review and Evaluation Committee (CREC) who studied cardiac dysfunction rates for patients receiving trastuzumab [3]. Their criteria for cardiac dysfunction included the presence of any of the following:
1) Cardiomyopathy characterized by the decrease in cardiac left ventricular ejection fraction (LVEF) that was either global or more severe in the septum;
2) Symptoms of congestive heart failure (CHF);
3) Associated signs of CHF, including but not limited to S3 gallop, tachycardia, or both; and
4) Decline in LVEF of at least 5% to less than 55% with accompanying signs or symptoms of CHF, or a decline in LVEF of at least 10% to below 55% without accompanying signs or symptoms.
The American College of Cardiology and the American Heart Association have developed a way to classify heart failure patients. Depending on the stage of the patient, management strategies change.
- Stage A patients are targeted to reduce risk factors that could lead to heart failure.
- Stage B patients have asymptomatic heart failure and are prescribed beta blockers and ACE-inhibitors to prevent further left ventricular remodeling.
- Stage C patients are provided diuretics, digoxin, and/or aldosterone antagonists in addition to therapies mentioned previously to further manage symptoms.
- Stage D patients have severe heart failure that is refractory to treatment. These patients may need to be evaluated for more invasive cardiac interventions (Table 1).
Table 1. American Heart Association (AHA) and American College of Cardiology (ACC) stages of heart failure
|
Stage
|
Description
|
|
A
|
Patients at high risk for developing heart failure
|
|
B
|
Asymptomatic heart failure
|
|
C
|
Symptomatic heart failure
|
|
D
|
Heart failure refractory to treatment
|
Another way to classify heart failure is by using the New York Heart Association’s classification to categorize patients according to limitations on physical activity.
- Class I patients generally do not have any restrictions with normal every day physical activity. These patients should participate in lifestyle modifications to reduce risk factors (proper diet, exercise, smoking cessation etc.).
- Class II patients may have slight restrictions with normal every day activities and will benefit from beta blockers and ace inhibitors.
- Class III patient will experience definite limitations with activity with dyspnea on exertion and orthopnea. These patients will benefit from dietary modifications and diuretics (in addition to the medications listed above) to control fluid retention and overload.
- Patients who are Class IV will have significant symptoms even at rest and will require more invasive intervention in addition to the medical therapies described above (Table 2).
Table 2. New York Heart Association (NYHA) clinical classification of heart failure
|
Class
|
Description
|
|
I
|
No restrictions with physical activity
|
|
II
|
Slight restrictions with normal everyday activities
|
|
III
|
Marked limitations with physical activity due to symptoms but no symptoms at rest
|
|
IV
|
Severe limitations with physical activity. May have symptoms at rest
|
Cardiotoxicity from chemotherapy depends on numerous factors that depend on both the characteristics of the patient and the chemotherapy itself. Some patient factors include age, underlying disease, previous history of cardiovascular disease, prior mediastinal radiation, and the presence of various modifiable cardiovascular risk factors [4]. Chemotherapeutic factors include the type of drug, potential drug-drug interactions, route of administration, cumulative dosage, and allergic effects [1-3]. Thus, it is important for all cancer patients to be properly evaluated to assess for baseline cardiovascular function prior to initiation of therapy.
Evaluation of cardiovascular function and assessment of potential modifiable cardiac risk factors is an important part of cancer management and can be initiated in the primary care setting. This begins with a comprehensive clinical exam to assess baseline function such as exercise capacity and resting heart rate [5]. Electrocardiograms (EKGs) can be useful in evaluating for conduction abnormalities, QT interval prolongations, and other underlying cardiac disease. Echocardiograms can determine baseline ejection fraction, systolic and diastolic cardiac function or dysfunction, and wall motion abnormalities, all of which may be worsened by chemotherapy and lead to arrhythmias and/or heart failure. Although informative, cardiac MRI and CT scans are not as useful to assess cardiac function prior to initiation of chemotherapy.
Cardiotoxicity as a result of chemotherapy can occur in the early, middle, or late progression of cancer. It can cause numerous cardio toxic pathology including hyper and hypotension, LV dysfunction, QT prolongation, arrhythmias, heart failure, and thromboembolism. The range of side effects can be from asymptomatic, temporary cardiac abnormalities to life threatening acute coronary syndromes, acutely decompensated heart failure, or even death [1-3]. However, because the initial changes in cardiac function can be subclinical and can occur at any time, it is important to maintain constant close monitoring of cardiac function throughout the treatment process.
Cardiovascular morbidity and mortality depends on the class of agent involved in therapy. The three broad categories of agents are implicated in cardiovascular toxicity are the anthracyclines, non-anthracyclines, and biological or targeted agents.
Anthracycline cancer chemotherapeutics include doxorubicin, daunorubicin, idarubicin, epirubicin, and mitoxantrone. Historically, these agents were limited by dose dependent myelosuppression, which prevented high dose therapy that could possibly lead to cardiotoxicity. Recent advancements in both chemotherapy and supportive management has led to higher doses being used, which has resulted in increased risk of side effects. As of now, anthracyclines are the class of chemotherapeutic agents most commonly associated with cardiotoxicity [4].
Doxorubicin interacts with DNA by binding to the enzyme Topoisomerase-II and stopping the replication of DNA. In the process, it generates toxic free oxygen radicals, leading to lipid peroxidation of membranes and replacement of myocytes by fibrous tissues. Topoisomerase-II alpha is overexpressed in tumors but its analog, Topoisomerase-II beta, is expressed in normal cardiac myocytes. Binding of doxorubicin to this subset of enzymes triggers the cardiac toxicity [6].
Cardiac function monitoring should occur prior to initiation of doxorubicin therapy [7]. Baseline radionuclide angiocardiography should be performed to calculate left ventricular ejection fraction (LVEF) and then a subsequent study should be done at least three weeks after the completion of one cycle, but prior to the next dosing cycle. Patients with a normal baseline LVEF of ≥50% should have additional angiocardiography performed based off of their underlying risk factors. A second study should be performed after 250-300 mg/m2 cumulative dose. In patients with known heart disease, abnormal EKG findings, concurrent cyclophosphamide therapy, or a history of radiation exposure, another study should be performed at 400mg/m2. If these risk factors are not present, the study can be done at 450 mg/m2. Doxorubicin therapy should be discontinued if the LVEF drops below 50% [7].
Patients with LVEF ≤50% require closer monitoring. In patients with EF between 30 and 50%, radionuclide angiocardiography should be performed prior to each dose. In patients with LVEF <30%, doxorubicin should not be started. Patients with a baseline LVEF ≤50% who experience a decrease in EF ≥10% or have a final EF ≤30% should stop doxorubicin therapy [7].
Clinical manifestations of anthracycline induced cardiomyopathy can occur anywhere within one year of initiation of chemotherapy, or up to 20 years afterwards, but the peak time to appearance of heart failure symptoms is within 3 months of finishing therapy [4]. Acutely, anthracycline infusion can cause a sudden decline in myocardial contractility, which may lead to arrhythmias, heart failure, or subclinical EKG changes. However, this acute change is usually reversible. Overall, acute cardiotoxicity is a rare event, but subclinical changes may occur, leading to cardiotoxicity many years after therapy has finished.
Chronic cardiotoxicity of anthracyclines may not be seen until 10-20 years after therapy has started. The most common presentation starts with systolic or diastolic cardiac dysfunction and eventually causes cardiomyopathy that leads to clinical heart failure due to the permanent, irreversible loss of cardiomyocytes. The incidence of cardiotoxicity depends on the cumulative dose used, with rates of 0.14% with cumulative doses <400 mg/m2, 7% between 400-550 mg/m2, and 18% at doses >700 mg/m2 [4].
The prognosis of anthracycline induced cardiotoxicity is directly related to the severity of symptoms. However, it is unclear if there is any benefit to prophylactically using medical therapy (such as beta blockers or ace inhibitors) prior to initiation of anthracycline therapy. In addition, it is unclear if management of anthracycline induced cardiac dysfunction should be addressed similarly to heart failure, arrhythmias, or coronary artery disease caused by other factors. Nevertheless, patients should receive optimal management from a team based approach according to current guidelines (Figure 1).
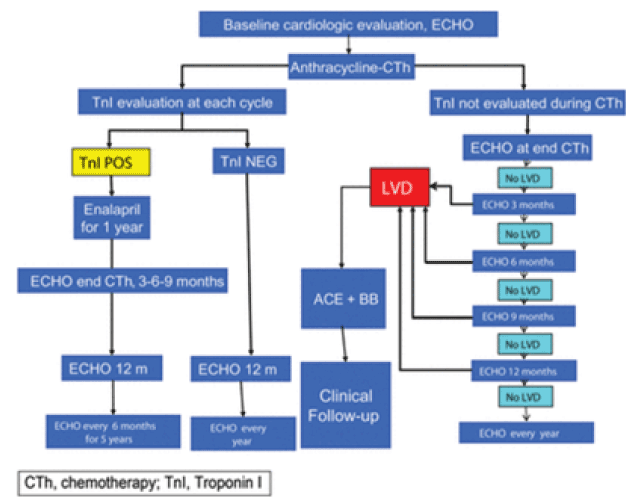
Figure 1. Cardiotoxicity management algorithm for patient receiving anthracyclines
Non-anthracyline agents can be broadly categorized into antimetabolites, microtubule agents, and alkylating agents. Within these classes, 5-fluorouracil (5-FU) of the antimetabolite class is the most common agent associated with cardiotoxicity, and is the second most common agent overall after the anthracyclines [8,9]. The overall incidence of cardiotoxicity of 5-FU is about 8% and it is most commonly associated with anginal pain that occurs with 72 hours of treatment initiation [8].
5-fluorouracil has multiple mechanisms of action, but predominantly as a thymidylate synthase inhibitor leading to decreased DNA synthesis and repair of cancer cells. However, the mechanism of cardiac pain and injury due to 5-FU is not fully understood and is based off a limited number of studies. However, coronary vasospasm due to transient vasoconstriction is the most likely cause. Several studies have shown that both the coronary artery and brachial artery exhibit vasospasm after 5-FU administration [10]. Other studies in animal models have shown a dose related vasospasm that resolves after 5-FU is stopped [11]. Other potential causes are endothelial damage and transient stress induced cardiomyopathies. In addition to chest pain, manifestations of 5-FU cardiotoxicity include subclinical yet asymptomatic EKG changes to acute pulmonary edema, arrhythmias, and myocardial infarctions with ST segment and cardiac biomarker elevations.
Multiple potential risk factors for cardiotoxicity secondary to fluorouracil have been reported. Some of these include age, underlying cardiovascular disease such as coronary artery disease and cardiomyopathy, history of radiation therapy, and usage of other medications that may be cardio toxic. However, most reports of cardiotoxicity in patients using fluorouracil occur in patients without any cardiovascular disease history, which shows that pre-existing cardiac disease is not predictive of cardio toxic risk.
The risk of cardiotoxicity of 5-FU depends on the route and schedule of administration [12]. In general, infusions have higher risk of cardiotoxicity in comparison to bolus regimens [8,9]. However, the relationship between the cumulative dosage and cardiotoxicity risk is unclear. However, if signs and symptoms of cardiotoxicity arise, fluorouracil therapy should be stopped immediately to assess whether the onset of symptoms correlate directly with the therapy involved. In general, re-challenging the patient should be avoided, although switching to a bolus regimen may be more beneficial In addition, patients should be provided with anti-anginal agents for symptomatic relief.
Other antimetabolites have similar cardio toxic profiles as 5-fluorouracil. Capecitabine in particular has a similar risk profile [13]. Fludarabine is not associated with cardiac toxicity as a single agent, but when combined with melphalan, can cause severe cardiac toxicity. Cytarabine can lead to pericarditis, pericardial effusion, and pericardial tamponade, which should be treated with high dose corticosteroids.
Her2 agents
HER-2 agents such as trastuzumab and pertuzumab are monoclonal antibodies that work against the HER-2 receptors. HER-2 receptors are involved in gene signaling and are important in stimulating cell proliferation. In certain types of breast cancers, these genes are over-expressed, allowing cancer cells to bypass the checkpoints of normal mitotic cell division, leading to uncontrolled cancer growth. Trastuzumab binds to the HER2/neu receptor to cause arrest of cells in the G1 phase of the cell cycle to restrict cellular growth and turnover.
The mechanism of trastuzumab leading to cardiotoxicity is not well understood [14-16]. However, the cardiotoxicity risk for trastuzumab is small to moderate compared to more toxic agents such as the anthracyclines and fluorouracil. In addition, the cardiotoxicity caused by trastuzumab is not dose dependent like other chemotherapeutic agent and is usually reversible. Anthracycline usage results in type I cardiac dysfunction which is associated with myocyte destruction. Trastuzumab however, is associated with Type II dysfunction, which leads to myocyte “stunning” as opposed to destruction, resulting in transient changes [17]. The manifestations of toxicity are more subtle, often seen with asymptomatic decrease in left ventricular ejection fraction and transient EKG changes.11,12,13 Acute decompensated heart failure does occur, but at lower rates than other agents [14-16].
Pertuzumab is a newer recombinant monoclonal antibody that is used in combination with trastuzumab as neoadjuvant therapy. It works by limiting the dimerization of the HER2 receptor with other HER receptors and is added to standard therapy because dual therapy against the HER2 receptor has shown to decrease disease progression and increase survival. The cardiotoxicity of pertuzumab is usually reported as a combination effect with trastuzumab. The addition of pertuzumab has not been shown to increase cardiac therapy when compared to trastuzumab therapy alone [18].
According to US FDA guidelines, patients treated with pertuzumab are recommended to have left ventricular ejection fractions evaluated at the beginning of treatment, and then at regular intervals afterwards [19]. This includes both individuals with metastatic disease (every 3 months) and those undergoing neoadjuvant treatment (every 6 weeks). If the LVEF drops below 45%, or is between 45-49% with 10% or greater drop in absolute value, pertuzumab (and trastuzumab) should be held and a repeat assessment should be performed within three weeks. At this point, the clinician should take into account the benefits versus the risks for the individual before deciding to reinitiate therapy.
Tyrosine kinase inhibitors
Tyrosine kinases are enzymes that activate other proteins through signal transduction. Lapatinib is a tyrosine kinase inhibitor that targets both the epidermal growth factor receptor (EGFR) and HER2/neu pathways. It attaches to tyrosine kinases to reversibly block ATP and block downstream signaling pathways. It is usually used as combination therapy with other HER2 agents for breast cancers that express HER2.
Lapatinib associated cardiotoxicity is generally reversible and non-progressive [20,21]. If present, it is associated with a symptomatic or asymptomatic decrease in left ventricular ejection fraction decreases (~20%) [21]. However, cardiotoxicity does not appear to be dose related and permanent myocardial muscle changes are rare. However, the overall Lapatinib related cardiotoxicity rates remain unclear.
Bevacizumab is a recombinant monoclonal antibody that blocks angiogenesis by inhibiting vascular endothelial growth factor A. The cardio toxic effects of Bevacizumab is due to the myocardium’s dependence on constant angiogenesis [22]. Although the occurrence of hypertension is a known side effect, the incidence of cardiotoxicity leading to heart failure or cardiomyopathy is low (<3%) [23]. However, this data is mostly collected from the adverse events of clinical trials involving breast cancer patients. Thus, the actual cardiotoxicity rates of Bevacizumab remain unknown (Table 3).
Table 3. Cardiovascular toxicity induced by chemotherapy, targeted agents, and radiotherapy: ESMO clinical practice guidelines
|
CV evaluation before anticancer treatment with potential nonreversible (Type I) or reversible (type II) cardiotoxicity
|
|
Guideline statements
|
Level of evidence
|
Grade of recommendation
|
|
Baseline evaluation
|
|
Patients undergoing chemotherapy should have careful clinical evaluation and assessment of CV risk factors and comorbidities. Strict attention should be paid to patient comorbidities, especially coronary artery disease and hypertension, in those patients receiving multitargeted agents, and these comorbidities should be robustly managed before and during therapy
|
I
|
A
|
|
Patients should be considered at risk for cardiac toxicity in case they have history of exposure to any of the following cumulative doses of anthracyclines as specified below.
- Doxorubicin >500 mg/m2
- Liposomal doxorubicin >900 mg/m2
- Epirubicin >720 mg/m2
- Mitoxantrone >120 mg/m2
- Idarubicin >90 mg/m2
|
I
|
A
|
|
LVEF assessment is mandatory for basal evaluation cardiac function before potential cardio toxic cancer treatment
|
I
|
A
|
|
should be recorded. The QT time A standard 12-lead ECG should be corrected for heart rate (QTc) with Bazett's formula (QTc = QT/√RR)
|
I
|
B
|
|
Echocardiography is the standard procedure for basal assessment of cardiac structure, performance and hemodynamics. Multiple gated acquisition (MUGA) scan can reduce interobserver variability with the disadvantages of including the exposure to radioactivity and the limited information than can be obtained on cardiac structure and diastolic function. Magnetic resonance imaging (MRI) is another method used to evaluate myocardial function. Its spatial resolution is higher than that of echocardiography, but its temporal resolution is lower.
|
I
|
A
|
|
Assessment by ultrasound should obtain 2D or 3D images in the left ventricular parasternal long- and short-axis views and in the apical four- and two-chamber long-axis views. For the analysis of diastolic function, the following parameters should be measured: the ratio of early peak flow velocity to atrial peak flow velocity (E/A ratio; normal value >1), the deceleration time of the early peak flow (DT; normal value <220 ms) and the isovolumic relaxation time (IVRT; normal value <100 ms). Left ventricular end-diastolic diameter (normal value, 47 ± 4 mm) should be measured to test for ventricular dilatation
|
I
|
A
|
|
Cardiac biomarkers such as the troponins and brain natriuretic peptides (BNP), and neutrophil glucosaminidase-associated lipocalin as a marker of renal injury, may be expected to be elevated with significant cardiotoxicity. Although it is not yet established whether their routine monitoring is useful in predicting cardiotoxicity, and this needs to be examined in prospective studies, there is a strong case to incorporate their use in the clinical trial setting
|
III
|
B
|
|
Treatment optimization of pre-existent cardiopathies: BB and ACE inhibitors where appropriate, maximize medical therapy for patients with coronary artery disease, coronary revascularization if clinically appropriate
|
I
|
A
|
|
To minimize cardiotoxicity, the use of liposome-encapsulated doxorubicin and the use of an appropriate cardioprotectant regimen (as dexrazoxane, BB, ACE-inhibitors, AT1-antagonists) should be considered and planned in all patients at high risk of cardiotoxicity
|
III
|
B
|
|
Cardiac monitoring
|
|
Patients receiving anthracyclines and/or trastuzumab in the adjuvant setting should perform serial monitoring of cardiac function at baseline, 3, 6 and 9 months during treatment, and then at 12 and 18 months after the initiation of treatment. Monitoring should be repeated during or following treatment as clinically indicated. Limited data are available for elderly patients: increased vigilance is recommended for patients ≥60 years old
|
I
|
A
|
|
Patients treated for metastatic disease: LVEF should be monitored at baseline and then infrequently in the absence of symptoms
|
II
|
A
|
|
Troponin I or BNP concentrations seem to identify patients at risk of cardiotoxicity, specifically in case of administration of type I agents (such as anthracyclines). Performing baseline assessment of biomarker concentrations and periodic measurements during therapy (every each cycle) may identify patients who need further cardiac assessment
|
III
|
B
|
|
Assessment of cardiac function is recommended 4 and 10 years after anthracycline therapy in patients who were treated at <15 years of age, or even at age >15 years but with cumulative dose of doxorubicin of >240 mg/m2or epirubicin >360 mg/m2
|
II
|
B
|
|
LVEF reduction of ≥15% from baseline with normal function (LFEV ≥ 50%) is an indication to continue anthracyclines and/or trastuzumab. LVEF decline to <50% during anthracyclines containing regimens necessitate reassessment after 3 weeks. If confirmed, hold chemotherapy, consider therapy for LVD and further frequent clinical and echocardiographic checks. In case of LVEF decline to <40% stop chemotherapy, discuss alternatives and treat LVD
|
II
|
B
|
2021 Copyright OAT. All rights reserv
|
LVEF decline to <50% during trastuzumab therapy (post-anthracyclines) necessitate reassessment after 3 weeks. If confirmed, continue trastuzumab and consider therapy for LVD and further frequent clinical and echocardiographic checks. In case of LVEF decline to <40% stop trastuzumab and treat LVD
|
|
|
|
Aggressive medical treatment of those patients, even asymptomatic, who show LVD at DEcho after anthracycline therapy is mandatory, especially if the neoplasia could have a long-term survival; it consists of ACE inhibitors and b-blockers and the earlier HF therapy is begun (within 2 months from the end of anthracycline therapy), the better the therapeutic response
|
|
|
- Floyd JD, Nguyen DT, Lobins RL, Bashir Q, Doll DC, et al. (2005) Cardiotoxicity of cancer therapy. J Clin Oncol 23: 7685. [Crossref]
- Monsuez JJ1, Charniot JC, Vignat N, Artigou JY (2010) Cardiac side-effects of cancer chemotherapy. Int J Cardiol 144: 3-15. [Crossref]
- Seidman A, Hudis C, Pierri MK, Shak S, Paton V, et al. (2002) Cardiac Dysfunction in the Trastuzumab Clinical Trials Experience. J Clin Oncol 20: 1215-1221. [Crossref]
- Von Hoff DD, Layard MW, Basa P, Davis HL Jr, Von Hoff AL, et al. (1979) Risk factors for doxorubicin-induced congestive heart failure. Ann Intern Med 91: 710-717. [Crossref]
- Curigliano G, Cardinale D, Suter T, Plataniotis G, de Azambuja E, et al. (2012) Cardiovascular toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidelines. Ann Oncol 23: vii155-vii166. [Crossref]
- Lyu YL, Kerrigan JE, Lin CP, Azarova AM, Tsai YC, et al. (2007) Topoisomerase IIbeta mediated DNA double-strand breaks: implications in doxorubicin cardiotoxicity and prevention by dexrazoxane. Cancer Res 67: 8839-8846. [Crossref]
- Schwartz RG, McKenzie WB, Alexander J, Sager P, D’Souza A, et al. (1987) Congestive heart failure and left ventricular dysfunction complicating doxorubicin therapy. Seven-year experience using serial radionuclide angiocardiography. Am J Med 82: 1109-1118. [Crossref]
- Labianca R, Beretta G, Clerici M, Fraschini P, Luporini G (1982) Cardiac toxicity of 5-fluorouracil: a study on 1083 patients. Tumori 68: 505-510. [Crossref]
- Anand AJ (1994) Fluorouracil cardiotoxicity. Ann Pharmacother 28: 374-378. [Crossref]
- Shoemaker LK, Arora U, Rocha Lima CM (2004) 5-fluorouracil-induced coronary vasospasm. Cancer Control 11: 46-49. [Crossref]
- Südhoff T, Enderle MD, Pahlke M, Petz C, Teschendorf C, et al. (2004) 5-Fluorouracil induces arterial vasocontractions. Ann Oncol 15: 661-664. [Crossref]
- Saif MW, Shah MM, Shah AR (2009) Fluoropyrimidine-associated cardiotoxicity: revisited. Expert Opin Drug Saf 8: 191-202. [Crossref]
- Saif MW, Quinn MG, Thomas RR, Ernst A, Grem JL (2003) Cardiac toxicity associated with capecitabine therapy. Acta Oncol 42: 342-344. [Crossref]
- Keefe DL (2002) Trastuzumab-associated cardiotoxicity. Cancer 95: 1592-1600. [Crossref]
- Perez EA, Rodeheffer R (2004) Clinical cardiac tolerability of trastuzumab. J Clin Oncol 22: 322-329. [Crossref]
- Fiúza M (2009) Cardiotoxicity associated with trastuzumab treatment of HER2+ breast cancer. Adv Ther 26 Suppl 1: S9-17. [Crossref]
- Ewer MS, Lippman SM (2005) Type II chemotherapy-related cardiac dysfunction: time to recognize a new entity. J Clin Oncol 23: 2900-2902. [Crossref]
- Swain SM, Ewer MS, Cortés J, Amadori D, Miles D, et al. (2013) Cardiac tolerability of pertuzumab plus trastuzumab plus docetaxel in patients with HER2-positive metastatic breast cancer in CLEOPATRA: a randomized, double-blind, placebo-controlled phase III study. Oncologist 18: 257-264. [Crossref]
- https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=17f85d17-ab71-4f5b-9fe3-0b8c822f69ff
- Sendur MA, Aksoy S, Altundag K (2013) Cardiotoxicity of novel HER2-targeted therapies. Curr Med Res Opin 29: 1015-1024. [Crossref]
- Perez EA, Koehler M, Byrne J, Preston AJ, Rappold E, et al. (2008) Cardiac safety of lapatinib: pooled analysis of 3689 patients enrolled in clinical trials. Mayo Clin Proc 83: 679-86. [Crossref]
- Varga ZV, Ferdinandy P2, Liaudet L3, Pacher P4 (2015) Drug-induced mitochondrial dysfunction and cardiotoxicity. Am J Physiol Heart Circ Physiol 309: H1453-1467. [crossref]
- Miller KD, Chap LI, Holmes FA, Cobleigh MA, Marcom PK, et al. (2005) Randomized phase III trial of capecitabine compared with bevacizumab plus capecitabine in patients with previously treated metastatic breast cancer. J Clin Oncol 23: 792-799. [Crossref]

