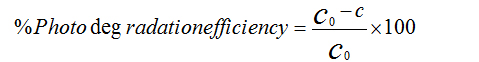Abstract
A sol-gel method was used to prepare four samples of Titanium dioxide TiO2 by different weight percent of tetrapropoyl ammonium hydroxide solution(TPAOH). The effect of different weight percent of TPAOH on the properties and efficiency of TiO2 was studied to degradation of methylene blue (MB) dye. The samples were characterized with X-ray diffraction(XRD),ultraviolet and visible spectroscopy (UV–Vis), Photoluminescence (Pl) emission spectra, surface area measurements (BET),and transmission electron microscopy (TEM). Furthermore, the photocatalytic performance of TiO2 was measured in the degradation of methylene blue (MB) dye. The results demonstrate that the best weight percent of TPAOH for the preparation of TiO2 nanoparticles at high properties and efficiency is 2.88 wt% .
Key words
Sol-gel, TiO2, Tetrapropoyl ammonium hydroxide, MB dye
Introduction
For the past ten years, the growth in population, development of technological, industrial and agricultural expansions have affected the environment and the health of human beings in society [1]. In the environment have been searching for suitable techniques of eliminating hazardous dye discharges that are released in the water bodies that result in the increase of dangerous environmental harms that have an effect on the health of human beings and the marine medium [2,3]. There have been many advances that have been incorporated to reduce the rate of toxicity caused by these dyes, methylene blue (MB) from the water [4], pinpoint these methods that include adsorption, especially on high plane area chains precipitation of chemicals, biological membranes, and ion-exchange processes and also through sedimentation [5]. These methods were considered to be effective in the removal of these chemical dyes although the processes were considered to be slow and there was the need to incorporate expensive materials in the removal of these dyes in the photocatalysis process. Also, these methods led to the researches being conducted on the of the sol-gel method in the synthesis of TiO2 nanoparticles in the process. Its relevance was also researched in the photocatalytic deprivation of methylene blue through the use of Titanium dioxide [6,7]. The synthesis of TiO2 nanoparticles by the sol-gel method is among the suggested techniques among the many techniques including the use of Cu2O that has been used widely in photocatalysis as a typical photoelectrode in the electrochemical cells especially under irradiation by visible light [8]. The splitting of water produced hydrogen fuel, although the fuel was not enough to be used in the process of combustion. This raised many debates in the research of suitable techniques of degradation of dyes such as methylene blue (MB) that were suggested in the removal of these effluent substances from our ecosystem [9]. However, the advantages attached to the use of TiO2 and that which is doped as photo-catalysts triggered the research in this field [10]. Latest studies have paid attention to the use of TiO2 based photocatalysts for the filth of dyes in the aqueous solutions. Nanocrystalline TiO2 is an oxide semiconductor material that had been studied in numerous experiments to develop a photocatalytic water treatment process that is efficient [11,12]. The process where TiO2 is used is characterized by high reaction rates and short periods of treatment that arise due to the quick oxidation processes caused by the hydroxyl radicals [13]. The study of these uses motivated the research and development of the several techniques employed in the preparation of catalysts that are used for different tasks in the degradation of methylene blue [14]. There is the need for the increase in the catalytic activity so as to enhance the competence of the whole process especially if the process is to be incorporated in the treatment of water in large scales [15]. The resourcefulness of TiO2 gives it a wide application in different areas of various products such as the manufacturing of pigments, development of electrochemical electrodes, in the production of toothpaste and capacitors [16]. Also, the oxidation process of the photocatalytic process where semiconductors such as TiO2, CdS, Cu2O, and ZnO has been widely studied in the past has been a point of concern in the degradation process of methylene blue (MB) [17]. This was due to the attention brought about by the hazardous organic substances released into the environment by the textile, technological and agricultural industries strive to produce products that are extensively needed across the globe [18,19]. This paper aims at synthesizing Titanium dioxide (TiO2) nanoparticles via the sol-gel method and evaluating the application of TiO2 to the photocatalytic degradation of the dye direct blue.
Experimental
Photocatalyst preparation
All chemicals in this experiment were analytical grade and used as received without further purification. Titanium dioxide TiO2 nanoparticles were prepared via sol-gel method according to the following route: different weight percent of tetrapropoyl ammonium hydroxide solution (TPAOH) (10%) (0.96, 1.44, 2.88, and 3.92% ) were added to the mixture of 5.28 ml distilled water, 16.8 ml ethanol (96%) and 0.05 ml nitric acid with constant stirring at room temperature for 1 hr. Then, 5 ml of Titanium(IV) butoxide (TBT) (97%), was added slowly with constant stirring at room temperature for another 1 hr. The produced white precipitate were collected by centrifugation, filtration, washing, and dried in a vacuum oven at 60°C for 24 hrs, crushed to fine powder followed and heated at 550°C for 5 h in muffle furnace.
Characterization techniques
X-ray diffraction (XRD) analysis was performed at room temperature with a Bruker axis D8, using Cu K radiation (λ= 1.540 A˚). The specific surface area was calculated from measurements Of N2 adsorption with a Nova 2000 series Chromatech apparatus at 77 K. Prior to measurement, all samples were treated for 2 h under a vacuum at 200°C. The band gap of the samples was identified using UV–Vis diffuse reflectance spectra (UV–Vis-DRS) in air at room temperature in the wavelength range of 200–800 nm, with an UV/Vis/NIR spectrophotometer (V-570, JASCO, Japan). Transmission electron microscopy (TEM) was recorded with a JEOL-JEM-1230 microscope, and the samples were prepared by suspending them in ethanol, thereafter ultrasonication for 30 min. placing a small amount of this solution onto a carbon-coated copper grid and dried the solution before loading the sample in the TEM.
Photocatalytic test
The photocatalytic activity test was studied by study degradation of methylene blue (MB) under UV light irradiation. The degradation reactions were carried out in a Pyrex glass beaker. The photocatalytic activity experiment was performed by adding 0.1 g of photocatalyst into the beaker with 300 ml aqueous methylene blue solution (50 ppm). This mixture was placed inside the photocatalytic reactor for 60 minutes and then O2 flow was bubbled through the reactor with constant flow rate to keep the mixture homogeneous. The reaction was initiated irradiation process when the UV-lamp was switched on. The experiment was performed at room temperature and pH about 7. The resulting mixture was taken after irradiation and the catalyst separated from solution by centrifugation process. After that, the content of methylene blue in the solution after irradiation was determined by monitor the absorbance of MB solutions under constant experimental conditions using UV-Vis spectrophotometer which was recorded on a Shimadzu UV-2450 with 1 cm (path length) quartz cuvette. The photodegradation efficiency of methylene blue was calculated by applying the following equation:
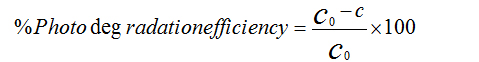
Where C0 is the original content of methylene blue and C is the residual methylene
blue in solution.
Results and discussion
Photocatalysts characterization
Phase analysis: TiO2 nanoparticles which prepared by different weight percents of tetrapropoyl ammonium hydroxide solution (TPAOH) were characterized by X-ray diffraction (XRD) analysis to determine their phase compositions. The X-ray diffraction patterns of TiO2 nanoparticles prepared by different weight percent of (TPAOH) were shown in Figure 1. The results reveal that TiO2 phase is present for all samples as a pure TiO2 phase. As the weight percents of tetrapropoyl ammonium hydroxide solution was increased, the diffraction peaks of the samples gradually became narrow and sharp. This change indicates that the particle size grew as weight percent of TPAOH increased.
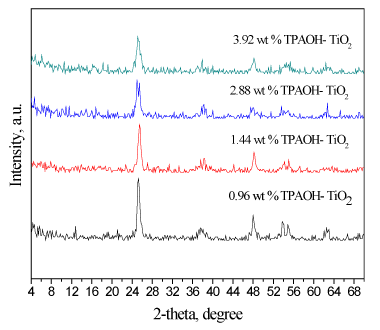
Figure 1. XRD patterns of as-synthesized TiO2 nanoparticles prepared at different weight percent of TPAOH.
Surface area analysis: The Specific surface area (SBET) of TiO2 nanoparticles which were prepared by different weight percent of tetrapropoyl ammonium hydroxide solution (TPAOH) such as 0.96%, 1.44% , 2.88% and 3.92% are tabulated in Table 1. The results reveal that the surface area of the TiO2 obtained at 2.88 wt% is 138 m2/g. As the weight percent increases, the surface area is increases from 80 m2/g at 0.96 wt% to 140 m2/g at 3.92 wt%. There was no significant change when adding 3.92 wt% of tetrapropoyl ammonium hydroxide solution. Thus, the TiO2 prepared by 2.88 wt% of tetrapropoyl ammonium hydroxide solution (TPAOH) is expected to have the highest photocatalytic activity, due to it has highest specific surface area which provide active sites to promote the adsorption capacity leading to higher photocatalytic activity.
Samples |
Surface area (m2/g) |
0.96 wt% TPAOH- TiO2 |
80 |
1.44 wt% TPAOH- TiO2 |
100 |
2.88 wt% TPAOH- TiO2 |
138 |
3.92 wt% TPAOH- TiO2 |
140 |
Table 1. BET surface area of TiO2 nanoparticles prepared at different weight percent of TPAOH.
Optical characterization
The study of UV–Vis radiation absorption constitutes an important agent for the evaluation of the changes produced in prepared nanoparticles by different treatments. The band gab of pure TiO2 phase is usually reported 3.2 eV. The UV-Vis diffuse reflectance spectra of TiO2 nanoparticles which prepared by different weight percent of tetrapropoyl ammonium hydroxide solution are show in Figure 2. The results reveal that the absorbance edges of the TiO2 samples regularly varied. As the weight percents of tetrapropoyl ammonium hydroxide solution (TPAOH) increased, the absorbance edges of the samples moved gradually to higher wavelength (shift on band gap to lower value), In UV-Vis/DR spectrum of samples the threshold edge of absorption band is observed in the range shorter than 400 nm in the area of the spectrum ultraviolet. The UV-Vis reflectance spectra, were used to calculate The band gap values of TiO2 samples by direct extrapolation the absorption edge by linear fit method with the onset corresponding wavelengths, using the following equation:
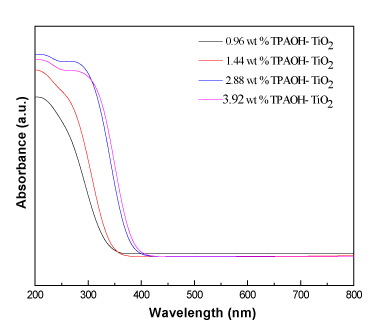
Figure 2. UV-Vis absorption spectra of TiO2 nanoparticles prepared at different weight percent of TPAOH.
E(ev) =1239.8/λ(nm)
The band gap value calculated for the prepared samples are demonstrated in Table 2. The results show that the band gap for TiO2 nanoparticles prepared by 0.96 wt%, 1.4 wt, 2.88 wt% and 3.92 wt% of tetrapropoyl ammonium hydroxide (TPAOH) are 3.54, 3.38, 3.20 and 3.12 eV, respectively, It is clear that the band gap decreased with increase wt% of tetrapropoyl ammonium hydroxide(TPAOH). So, the weight percent plays an important role in determining the band gap of the samplesTiO2.
Samples |
Band gap, eV |
0.96 wt% TPAOH- TiO2 |
3.54 |
1.44 wt% TPAOH- TiO2 |
3.38 |
2.88 wt% TPAOH- TiO2 |
3.20 |
3.92 wt% TPAOH- TiO2 |
3.12 |
Table 2. Band gap of TiO2 nanoparticles prepared by different weight percent of TPAOH.
Pl emission spectra were measured for samples excited at 265 nm at room temperature . Pl spectra were used to study the transfer of photogenerated electrons and holes and understand the separation and recombination of photogenerated charge carriers in the samples. Pl emission spectra is shown in Figure 3. The results reveal that Pl intensity decreased by increasing the weight percent of TPAOH. Also, we noticed that the position of emission spectra of TiO2 was shifted to longer wavelength as weight percent of TPAOH was increased, which are agree with UV-Vis results. The band gap estimated from PL emission spectra are very close to that estimated from UV-Vis spectra , indicating a decrease in the band gap energy and an increase in the amount of photogenerated electrons and holes available to contribute in photocatalytic reactions.
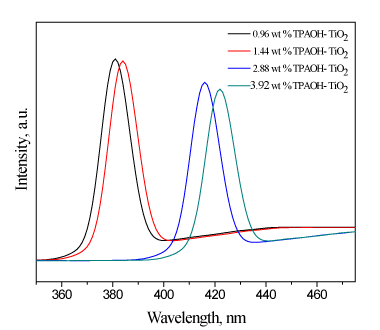
Figure 3. Pl spectra of TiO2 nanoparticles prepared at different weight percent of TPAOH.
TEM analysis
Figure 4 shows TEM images of TiO2 nanoparticles, prepared by different weight percent of TPAOH. The results demonstrated that the shape of TiO2 samples is a spherical shape. On increasing the weight percent of tetrapropoyl ammonium hydroxide from 0.96% to 1.44% increase the overall particle size of the produced nanoparticles. After increase in weight percent to 2.88% , there was decrease in particle size. Again, When weight percent is increase to 3.92% there was increase in particle size.
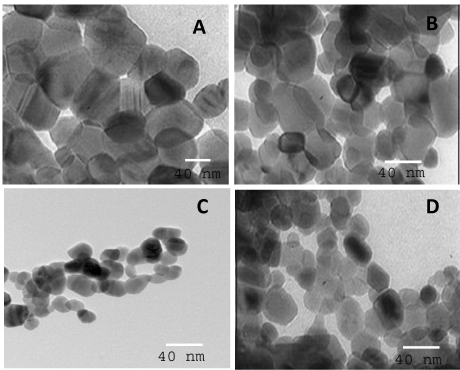
Figure 4. TEM images of TiO2 prepared at different weight percent of TPAOH, where, weight percent of TPAOH is 0.96 (A), 1.44 (B), 2.88(C) and 3.92(D).
Photocatalytic oxidation of MB-dye using Tio2 nanoparticles under UV irradiation light
The photocatalytic activity of TiO2 nanoparticles prepared by different weight percent of tetrapropoyl ammonium were tested using solution of methylene blue (MB) under UV light irradiation after 60 minutes are shown in Figure 5. The results reveal that as increase wt% of TPAOH from 0.96 to 3.92%, increase photocatalytic activity from 69 to 100%, respectively. However, increase wt% of TPAOH above 2.88%, i.e., 3.92%, has no significant effect on photocatalytic activity. Therefore, the optimum wt% of TPAOH is 2.88 wt% at which photocatalytic activity is 99%.
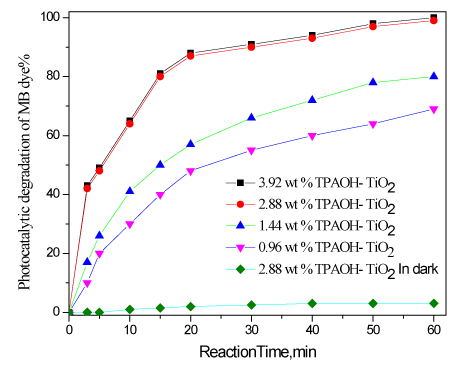
Figure 5. Photocatalytic degradation of TiO2 nanoparticles prepared by different weight percent of TPAOH.
Conclusions
A sol-gel method was used to prepare four samples of titanium dioxide TiO2 by different concentrations of tetrapropoyl ammonium hydroxide solution. In UV-Vis/DR spectrum of samples the threshold edge of absorption band is observed in the range shorter than 400 nm in the area of the spectrum ultraviolet. The highest wavelength of the TiO2 prepared by 2.88 wt% of TPAOH is 386.85 nm (shift on band gap to lower value), and It has lowest PL intensity , which means it has low rate of recombination of photogenerated electron- hole pairs. Therefore, the weight percent of TPAOH plays an important role in determining properties and efficiency samples of Titanium dioxide. Measurements of the photocatalytic degradation of methylene blue (MB) dye reveal that TiO2 prepared by 2.88 wt% of TPAOH exhibited the highest photocatalytic activity and active photocatalytic properties for water purification and may find potential applications in related fields.
References
- Herna´ndez-Rami´rez A, Medina-Rami´rez I (2014) Photocatalytic semiconductors: Synthesis, characterization, and environmental applications. Springer.
2021 Copyright OAT. All rights reserv
- Alca^ntara ACS, Kalia S (2015) Organic-inorganic hybrid nanomaterials. Berlin: Springer.
- Hussein AK (2015)Applications of nanotechnology in renewable energies—A comprehensive overview and understanding. Renewable and Sustainable Energy Reviews 42: 460-476
- Morales-Torres S, Pastrana-Martínez LM (2014) Nanostructured carbon–TiO2 photocatalysts for water purification: an overview.
- Zhao Z, Sun ZC, Zhao HF, Zheng M, Du P, et al. (2012) Phase control of hierarchically structured mesoporousanatase TiO2 microspheres covered with facets. J Mater Chem 22: 21965–21971.
- Ding X, Zhou S, Jiang L, Yang H ( 2011) Preparation, photocatalyticactivity and mechanism of nano-Titania/Nafion hybrid membrane. J Sol-Gel Sci Technol 58: 345-354.
- Chen X, Mao SS (2007) Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem Rev 107: 2891-2959. [Crossref]
- Lu X, Song C, Jia S, Tong Z, Tang X, Teng Y (2015) Low-temperature selective catalytic reduction of NOxwith NH3 over cerium and manganese oxides supported on TiO2–graphene. Chem Eng J 260: 776–784.
- Liu Y, Yang Q, Wei J, Xiong R, Pan C. X, Shi J (2011)Synthesis and photocatalytic activity of hydroxyapatite modifiednitrogen-doped TiO2. Mater Chem Phys 129: 654-659.
- Lonnen J, Kilvington S, Kehoe SC, Al-Touati F, McGuigan KG (2005) Solar and photocatalytic disinfection of protozoan, fungal and bacterial microbes in drinking water. Water Res 39: 877-883. [Crossref]
- Li D, Haneda H (2003) Synthesis of nitrogen-containing ZnO powdersby spray pyrolysis and their visible-light photocatalysis in gasphaseacetaldehyde decomposition. J Photochem Photobiol A 155: 171-178.
- Kamat PV, Meisel D (2002) Nanoparticles in advanced oxidation processes. Curr Opin Colloid Interface Sci 7: 282–7.
- Daghrir R, Drogui P, Robert D (2012) Photoelectrocatalytic technologies for environmental applications. A Journal of Photochemistry and Photobiology. Chemistry 238: 41-52.
- Han H, Ba R (2009) Buoyant photocatalyst with greatly enhanced visible-light activityprepared through a low temperature hydrothermalmethod. Ind EngChem Res 48: 2891–2898
- Herna´ndez-Rami´rez A, Medina-Rami´rez I (2014). Photocatalytic semiconductors: Synthesis, characterization, and environmental applications. Springer.
- Diallo MST, Fromer NA, Jhon MS (2014) Nanotechnology for sustainable development.
- Govindaraj R, Pandian MS, Ramasamy P, Mukhopadhyay S (2015) Sol–gel synthesized mesoporous anatase titanium dioxide nanoparticles for dye sensitized solar cell (DSSC) applications. Bulletin of Materials Science 38: 291-296.
- Dawson G, Chen W, Zhang T, et al.( 2010) A study on the effect of startingmaterial phase on the production of trititanate nanotubes. Solid State Sci 12: 2170–2176
- Aparicio M, Jitianu A, Klein LC (2012)Sol-Gel processing for conventional and alternative energy. New York: Springer.