The evaluation of blood pressure (BP) response allows us to estimate the left ventricular (LV) performance during physical effort. Under normal conditions, during the exercise (treadmill) test, systolic blood pressure (SBP) rises with increasing intensity of the applied work (usually up to 220 mmHg) and diastolic blood pressure (DBP) remains constant or oscillates about 10 mmHg. Conceptualizes be a hypertensive response to stress the finding of SBP >220 mmHg and/or elevation of 15 mmHg or more of DBP, starting with normal BP values at rest. It is suggested that individuals with hyperactive response across from the effort have future likelihood four to five times as high for become hypertensive, in relation those with normal curves of BP [1-3]. Interference of exercise on heart rhythm is by neuro-humoral changes represented by increased adrenergic tone, with reduction of vagal influence, electrophysiological changes, especially those which modify the electrical properties of driving cells and formation of cardiac stimulation and through hemodynamic rebalancing determined by BP changes, heart rate, myocardial oxygen consumption, inotropic changes, and others [4,5]. These variables, in special the rise of adrenergic tone, may induce the trigger of atrial fibrillation (AF) during the exercise. Men generally develop AF more frequently than do women. By 40 years of age, the lifetime risk of developing AF is almost 25% [6].
Pokushalov and colleagues [7] recently reported that renal sympathetic denervation (RSD) diminishes systolic and diastolic blood pressure in drug-resistant hypertensive patients and reduces AF recurrences when combined with pulmonary vein isolation. A strategy using percutaneous catheter-based delivery of radiofrequency (RF) energy was recently settled to interject the sympathetic innervation of the kidneys. This new procedure exposed no severe vascular or renal complications in the long term (up to 36 months). Our group believes that patients without hypertension, however presenting a hypertensive response associate to AF development during the treadmill test can be benefited if RSD be performed, reducing the adrenergic tonus, and consequently diminishing the increase in BP and the development of AF episodes.
This prospective longitudinal study involved 29 patients without hypertension and a family history AF. The study was conducted in agreement with the Helsinki declaration and approved by the ethics committee of our institution. All patients signed the informed consent term before inclusion. This study was conducted at the Hospital e Clínica São Gonçalo, Rio de Janeiro, Brazil. Patients were recruited from January to August 2015 from the Arrhythmias and Artificial Cardiac Pacing Service of the same hospital. Patients with the combination of the following criteria were consecutively enrolled: (i) mean 24-hour systolic ambulatory blood pressure measurements (ABPM) of <130 mmHg and mean 24-hour diastolic ambulatory blood pressure <80 mmHg, presenting a hypertensive response at the treadmill test; (ii) a physically normal heart with an ejection fraction of >50% as measured by echocardiography (Simpson’s method); (iii) no complaint of palpitations or arrhythmias record; (iv) age of 18 to 80 years, (v) estimated glomerular filtration rate (eGFR) >60 mL/min/1.73 m2 estimated by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation [8] (without microalbuminuria), and (vi) the capacity to read, comprehend, and sign the informed consent form and attend the clinical tests. The patients that presented any of the subsequent criteria were excluded: (i) pregnancy; (ii) valvular disease with significant adverse sequelae; (iii) unstable angina, myocardial infarction, transient ischemic attack or stroke within the 6 months before the procedure; (iv) renovascular abnormalities; (v) psychiatric disease; (vi) allergy to ionic contrast medium; (vii) the inability to be monitored clinically after the procedure; (viii) a known addiction to drugs or alcohol that affects the intellect; (ix) a serious health condition that, in the investigator opinion’s, may adversely affect the safety and/or efficacy of the participant or the study; (x) congestive heart failure presenting functional class II to IV symptoms according to New York Heart Association; (xi) Indexed left atrial (LA) volume, (LAD) >30 mL/m2 on transthoracic echocardiography; (xii) or a previous AF ablation record.
At baseline, the 19 subjects from the control group and the 10 patients from the RSD group underwent treadmill test, echocardiogram, blood and urine samples. At the 6th month of follow-up the 10 patients underwent RSD were submitted to an echo-Doppler of renal arteries, without a present, any complication and the exercise treadmill test [9] was repeated in both groups. The primary goal of this study evaluated if RSD can reduce hypertensive response on tread mill test. The RSD procedure has been described in detail previously [10]. The patients remained hospitalized in the ward for 24 h after the procedure.
The results are expressed as a mean and standard deviation for normally distributed data and as median with interquartile range otherwise. All statistical tests were two-sided. Comparisons between two-paired values were performed with the paired t-test in cases of a Gaussian distribution and by the Wilcoxon test otherwise. Comparisons between more than two-paired values were made by repeated-measures analysis of variance or by Kruskal–Wallis analysis of variance as appropriate, complemented by a post-hoc test. Categorical variables were compared with Fisher’s exact test. A P-value <0.05 was considered significant. Correlations between two variables were performed by Pearson’s chi-square test in case of a Gaussian distribution and with the Spearman correlation test otherwise. All statistical analyses were performed using the program Graphpad Prism v 7.0 (Graphpad Software, La Jolla, CA, USA).
The general features of both groups of patients are listed in Table 1. The variation (∆) between rest and exercise in SBP at baseline in control vs. RSD group was 87.3 ± 18.7 vs. 90.6 ± 19.5 mmHg, respectively (P=0.6553), and 87.3 ± 21.4 mmHg vs. 33.4 ± 11.5 mmHg, respectively (P<0.0001). When we compared baseline vs. the 6th month, no change occurred in the control group (P>0.9999) and the ∆ between rest and exercise in SBP in the RSD group was significant reduced (P<0.0001), as shown in Figure 1A. The same phenomenon was noted in DBP. The variation (∆) between rest and exercise in DBP at baseline in control vs. RSD group was 33.6 ± 13.6 vs. 34.1 ± 15.1 mmHg, respectively (P=0.9328), and 31.8 ± 13.6 mmHg vs. 12.4 ± 5.2 mmHg, respectively (P=0.0002). When we compared baseline vs. the 6th month, no change occurred in the control group (P=0.6869) and the ∆ between rest and exercise in DBP in the RSD group was significant reduced (P<0.0004), as shown in Figure 1B. The correlation between the increase in SBP and AF occurrence at baseline for control group was (by Pearson): r=0.9640; 95%CI: 0.9068 - 0.9863; P<0.0001 and for RSD group: r=0.9201; 95%CI: 0.6906 - 0.9813; P=0.0002. And at the 6th month of follow-up it was: r=0.9215; 95%CI: 0.8036 - 0.9698; P<0.0001 for control group, and r=0.4956; 95%CI: -0.1948 - 0.8576; P=0.1452 for the RSD group. The correlation between the increase in DBP and AF occurrence at baseline for control group was (by Pearson): r=0.7957; 95%CI: 0.5348 - 0.9181; P<0.0001 and for RSD group: r=0.9507; 95%CI: 0.7998 - 0.9886; P<0.0001. And at the 6th month of follow-up it was: r=0.7711; 95%CI: 0.4876 - 0.9075; P=0.0001 for control group, and r= -0.4468; 95%CI: -0.8401- 0.2543; P=0.1954 for the RSD group.
Table 1. General features of patients at baseline
|
Controls |
RSD |
P value |
N |
19 |
10 |
--- |
Age, years |
45.6 ± 6.0 |
43.2 ± 5.4 |
0.2995 |
Body mass index, kg/m2 |
26.8 ± 3.1 |
27.4 ± 4.5 |
0.6754 |
Male sex (%) |
13 (68%) |
9 (80%) |
0.6749 |
White ethnicity (%) |
16 (84%) |
7 (70%) |
0.6328 |
Type 2 Diabetes Mellitus (%) |
4 (21%) |
3 (30%) |
0.6647 |
Coronary artery disease |
6 (32%) |
1 (10%) |
0.3667 |
Hypertension |
0 (0%) |
0 (0%) |
--- |
Creatinine, mg/dL |
0.92 ± 0.10 |
0.89 ± 0.24 |
0.6369 |
eGFR, mL/min/1.73m² (CKD-EPI) |
99.3 ± 9.0 |
102.6 ± 12.5 |
0.4193 |
ACR, mg/g |
9.8 ± 5.4 |
11.0 ± 6.5 |
0.6001 |
Mean 24-hour ABPM, mmHg |
122.0 ± 6.8/73.4 ± 2.8 |
120.1 ± 7.9/72.1 ± 4.7 |
0.5043/0.3567 |
Echocardiographic parameters |
|
|
|
Indexed LV mass/BSA, g/m2 |
81.4 ± 6.7 |
83.5 ± 5.0 |
0.3925 |
LVEF, % (Simpson) |
66.3 ± 5.8 |
67.2 ± 6.0 |
0.6977 |
LVEDD, mm |
50.6 ± 2.2 |
52.0 ± 3.8 |
0.2171 |
LVESD, mm |
38.3 ± 3.1 |
37.6 ± 2.4 |
0.5399 |
Indexed LA volume, mL/m2 |
28.3 ± 1.4 |
27.5 ± 2.0 |
0.2184 |
Values are presented as Mean ± SD or %; ABPM, ambulatory blood pressure measurements; ACR, albumin: creatinine ratio; BSA, body surface area; EF, ejection fraction; eGFR, estimated glomerular filtration rate; LA, left atrium; LV, left ventricular; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; N, number of patients.
The Receiver Operating Characteristic (ROC) curves from the increase of SBP and DBP were area under the rock curve: 0.9847, 95%CI: 0.9540 – 0.9997, P<0.0001, sensitivity: 97%, specificity: 100%, cutoff point of ∆ SBP to trigger AF during treadmill test: >78 mmHg, and area under the rock curve: 0.8851, 95%CI: 0.7756 – 0.9945, P<0.0001, sensitivity: 94%, specificity: 91%, cutoff point of ∆ DBP to trigger AF during treadmill test: >28 mmHg, respectively. The variation (∆) between rest and exercise in metabolic equivalents (METs) reached at baseline in control vs. RSD group was 5.8 ± 1.7 vs. 5.1 ± 1.0 METs, respectively (P=0.2416), and 5.8 ± 1.8 vs. 8.4 ± 1.4 METs, respectively (P=0.0004). The comparison between baseline vs. the 6th month showed no change in the control group (P=0.9848) and the ∆ between rest and exercise in METs in the RSD group was significant augmented (P<0.0001), as shown in Figure 1C.
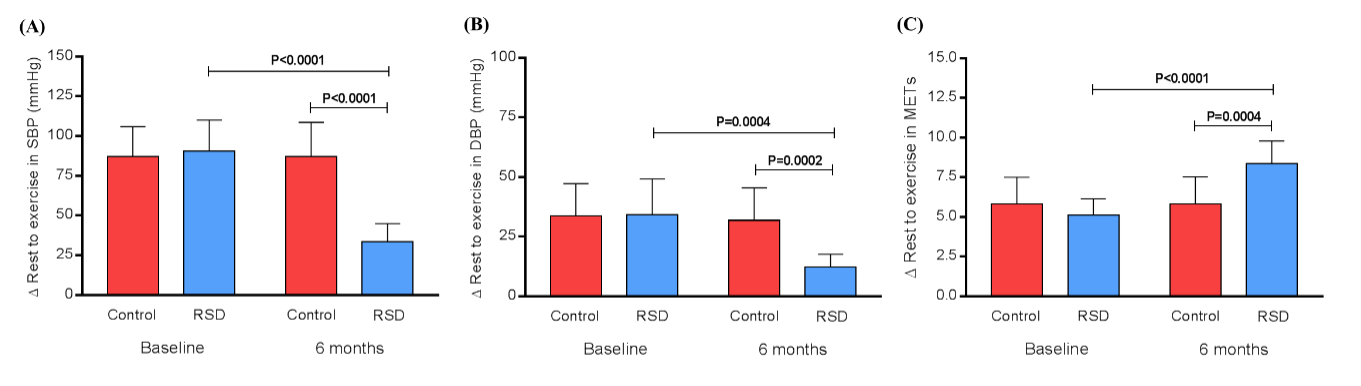
Figure 1. The variation (∆) between rest and exercise in SBP (A), DBP (B) and METs (C) at baseline and at the 6th month in control vs. RSD group. DBP, diastolic blood pressure; METs, metabolic equivalents; RSD, renal sympathetic denervation; SBP, systolic blood pressure; n=19 for the control group and n=10 for the RSD group.
In conclusion, our study shows that the hyperactive response in SBP and DBP is associated with AF occurrence in non-hypertensive patients, and after RSD there is a significant reduction in the hypertensive response to stress and reduction in AF events in this group.
None declared.
This study was funded by Pacemed (US $100,000), Rio de Janeiro, Brazil.
The authors are grateful to all participants included in this study. The authors also thank Pacemed for stimulating the development of this study and for providing technical support.
- Singh JP, Larson MG, Manolio TA, O'Donnell CJ, Lauer M, et al. (1999) Blood pressure response during treadmill testing as a risk factor for new-onset hypertension. The Framingham heart study. Circulation 99: 1831-1836. [Crossref]
- McHam SA, Marwick TH, Pashkow FJ, Lauer MS (1999) Delayed systolic blood pressure recovery after graded exercise: an independent correlate of angiographic coronary disease. J Am Coll Cardiol 34: 754-759. [Crossref]
- Allison TG, Cordeiro MA, Miller TD, Daida H, Squires RW, et al. (1999) Prognostic significance of exercise-induced systemic hypertension in healthy subjects. Am J Cardiol 83: 371-375. [Crossref]
- Hauswirth O, Noble D, Tsien RW (1968) Adrenaline: mechanism of action on the pacemaker potential in cardiac Purkinje fibers. Science 162: 916-917. [Crossref]
- Wit AL, Cranefield PF (1976) Triggered activity in cardiac muscle fibers of the simian mitral valve. Circ Res 38: 85-98. [Crossref]
- Lloyd-Jones DM, Wang TJ, Leip EP, Larson MG, Levy D, et al. (2004) Lifetime risk for development of atrial fibrillation: the Framingham Heart Study. Circulation 110: 1042-1046. [Crossref]
- Pokushalov E, Romanov A, Corbucci G, Artyomenko S, Baranova V, et al. (2012) A randomized comparison of pulmonary vein isolation with versus without concomitant renal artery denervation in patients with refractory symptomatic atrial fibrillation and resistant hypertension. J Am Coll Cardiol 60: 1163-1170. [Crossref]
- Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF III, et al. (2009) CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration): A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612.
- Carreira MA, Nogueira AB, Pena FM, Kiuchi MG2021 Copyright OAT. All rights reservon of Autonomic Dysfunction in Hemodialysis Patients Using the Exercise Treadmill Test: The Role of the Chronotropic Index, Heart Rate Recovery, and R-R Variability. PLoS One 10: e0128123.
- Kiuchi MG, E Silva GR, Paz LM, Chen S, Souto GL (2016) Proof of concept study: renal sympathetic denervation for treatment of polymorphic premature ventricular complexes. J Interv Card Electrophysiol. 2016 May 30. [Epub ahead of print]

