Purpose: Nosocomial infections are some of the main causes of worsening outcomes and death in aneurysmal subarachnoid hemorrhage patients. We hypothesize that combination of clinical parameters and blood biomarkers could increase the capacity of individual markers to dichotomize the patients at risk of infections.
Methods: The present study included 104 patients (69 infected/35 non-infected) from two independent European cohorts. Accuracy of biomarkers (serum amyloid A, C-reactive protein, neopterin and WBC) and clinical parameters (WFNS, GCS and age) were evaluated at hospital admission using receiver operating characteristic curves. The most accurate panel combination was obtained using Panelomix.
Results: At hospital admission, the most sensitive parameters for the stratification of patients at risk of developing an infection were SAA and the WFNS. To reach a SP of 100% (95% CI, 100–100), SE values of 26.9% (95% CI, 15.9–38.1) and 31.9% (95% CI, 21.7–43.5) were obtained respectively. Moreover, the combination of SAA, WBC, WFNS, and age significantly improved the SE to 64.3% (95% CI, 50–78.6).
Conclusions: At hospital admission the panel SAA, WBC, WFNS, and age appear as a promising tool for predicting in-hospital infections, which could lead to a better management of patients and in their associated outcomes.
Abbreviations: aSAH: aneurysmal subarachnoid hemorrhage; AUC: area under the ROC curve; CI: confidence interval; CRP: C-reactive protein; ELISA: enzyme-linked immunosorbent assay; GCS: Glasgow Coma Scale; GOS: Glasgow Outcome Scale; ICU: intensive care unit; MSD: mesoscale discovery; NP: neopterin; ROC: receiver operating characteristic; SAA: serum amyloid A; SE: sensitivity; SP: specificity; UTI: urinary tract infection; WBC: white blood cells; WFNS: World Federation of Neurosurgical Societies
Nosocomial infections are common complications in patients suffering from aneurysmal subarachnoid hemorrhage (aSAH). Pneumonia and urinary tract infections (UTI) are the most prevalent, developing in at least one third of aSAH patients [1,2]. Such infections can significantly impair patients’ outcomes, prolonging hospital stays, and increasing associated rates of morbidity and mortality [3-5]. Identifying patients at a higher risk of developing an infection is crucial to promptly improve treatment for a potentially long-lasting disability [6,7].
Blood biomarkers can be effective risk assessment indicators; they can provide information about the disease’s pathophysiology and, furthermore, they are objectively measurable throughout hospitalization [8,9]. In stroke patients, C-reactive protein (CRP) or white blood cells (WBC) are the two most commonly used infection markers [10,11]. However, for aSAH patients, no blood biomarkers are available in clinical practice. We recently showed that neopterin (NP) and serum amyloid A (SAA) could be promising predictive markers of infection as their plasma concentrations were significantly higher in patients developing infection during hospitalization compared to those who did not [12-14].
The utility of combining multiple parameters to improve the performance of individual markers has been described previously [15-19]. Therefore, in the present study, we postulated that combining previously described blood biomarkers with aSAH clinical parameters might be an efficient strategy for the stratification of patients in need of prophylactic antibiotherapy. Firstly, we evaluated how effectively different blood biomarkers (SAA, CRP, NP, and WBC) and clinical parameters (GCS, WNFS, Fisher, and age) at hospital admission were in identifying patients who subsequently developed an infection during hospitalization. Secondly, we tested which combination of parameters formed the best test panel for predicting the risk of infection.
Patient description
The present study included 104 patients from two different cohorts of patients: 25 patients admitted consecutively to the Charité Universitätsmedizin Hospital in Berlin (Germany) between January 2010 and August 2012 and 79 patients admitted consecutively to the Pitié-Salpêtrière Hospital in Paris (France) between July 2004 and April 2008.
The present study was approved by both hospitals’ ethics committees: the Local Research Ethics Committee at Charité Virchow Medical Center and the Patient Protection Committee at the Pitié-Sapêtrière Hospital respectively. As per the Declaration of Helsinki, all the patients or the legal representatives signed an informed consent form.
The inclusion criteria for the present study were: 1) aged 18 years old or above, 2) aSAH confirmed by computed tomography and angiography, and 3) hospital admission within 48 hours of the onset of the hemorrhage. Patients presenting with more than one hemorrhagic event, not enough sample volume, and/or missing clinical information were excluded (Supplementary figure 1).
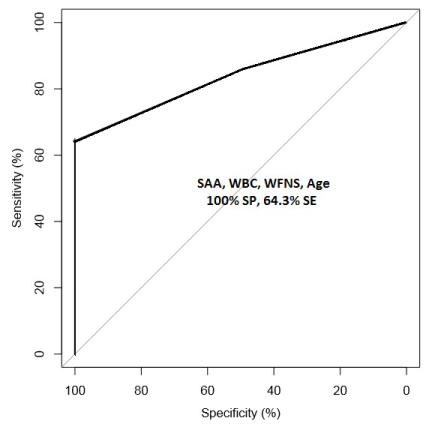
Figure 1. ROC curve of SAA, WBC, WFNS, and age test panel for the discrimination between infected and non-infected patients at hospital admission.
The severity of aSAH patients was assessed at hospital admission using the World Federation of Neurosurgical Societies scale (WFNS 1–5) [20]. Patients were dichotomized between good (WFNS 1–2) or poor neurological state (WFNS 3–5) depending on their initial clinical state. According to the Glasgow Coma Scale (GCS) patients were separated into three groups: patients with severe brain injury (GCS<9), moderate brain injury (GCS 9–12), and minor brain injury (GCS ≥ 13) [20,21]. The severity of the hemorrhage was established according to the Fisher score [22]: grade 1, no subarachnoid blood; grade 2, broad diffusion of subarachnoid blood; grade 3, with clots or thick layers of blood; grade 4, intraventricular hemorrhage or intracerebral hematoma, no clot; grade 5, intraventricular hemorrhage or intracerebral hematoma with clot.
The best type of intervention (clipping or coiling) was determined based on the location and size of each aneurysm.
The Glasgow Outcome Scale (GOS) was used to evaluate patients outcome one year after hospital admission. Depending on the level of functional dependence, patients were dichotomized into poor (GOS score of 1, 2, or 3) or good outcome groups (GOS score of 4 or 5) [23].
Infection status was established according to the International Sepsis Forum Consensus Conference on Definitions of Infection in the ICU [24]. Patients were classified as having or not this kind of infections: pneumonia, UTI or bloodstream infection. Bacteriological samples were taken at this time point, and the treatment was adjusted once results were obtained.
The demographic characteristics of the patients are shown in Table 1.
Marker measurement
Plasma samples were collected from patients on the day of hospital admission.
Concentrations of SAA, CRP, NP, and WBC were measured in these plasma samples collected on the day of hospital admission.
The Meso Scale Discovery (MSD) Vascular Injury Panel-I ECL assay was used to determine the levels of SAA and CRP in plasma, as per manufacturer’s instructions (MSD, Gaithersburg, MD). Plasma samples were diluted 1:2000, and analyte concentrations were determined using an electrochemiluminescence detection system using multi-array technology (SECTOR Imager 2400, Meso Scale Discovery) [25].
NP levels in plasma were measured using a commercial ELISA kit, as per manufacturer’s instructions (Brahms Gmbh, Hennigsdorf, Germany). Samples were diluted 1:4.
WBC count was determined using four-color flow cytometry on a FACSCalibur using CellQuest Software (BD Biosciences) [26].
All analytes were measured in duplicates (CV < 15%). Marker concentrations in the two different cohorts were normalized according to the median concentrations in each population set [27].
Statistical data analysis
Statistical analyses were performed using SPSS software (version 21, SPSS Inc., Chicago, IL). As the levels of the different analytes were not normally distributed, Mann-Whitney U-test was used for statistical comparisons between two unpaired groups. Fisher’s exact test and the Chi-squared test were used to assess whether the patients with and without infection were significantly different according to their gender, WFNS score, GCS, modified Fisher score, and outcome.
All statistical tests were bilateral, and a p-value <0.05 was considered statistically significant.
At hospital admission, receiver operating characteristic (ROC) curves were calculated for SAA, CRP, NP, and WBC as well as for the clinical parameters (GCS, WFNS, Fisher, and age). For each marker the SP value was restricted to 90-100% and the cut-off value was selected, in order to correctly classify 9 out of 10 patients that will not develop an infection. The pROC package for S+ (version 8.1., TIBCO Software Inc.) was used to calculate the values of areas under the partial area under the curve (AUC), specificity (SP), sensitivity (SE), and 95% confidence intervals (95% CI) [28].
Panel selection was performed using the PanelomiX tool, as previously described [15]. Briefly, the optimized cut-off values were obtained by modified iterative permutation-response calculations (rule-induction-like, RIL) using all the individual parameters, the different analytes (SAA, CRP, NP, WBC) as well as the different clinical parameters (GCS, WFNS, Fisher, age). The cut-off values of each molecule or clinical parameter were changed iteratively by 2% increment quantiles. After each iteration the SE value was calculated using an SP value set between 90%–100%. The panel size was limited to a maximum of four parameters
The demographic characteristics of the 104 patients included in the study are shown in Table 1. Most of the patients suffering from aSAH were women, however, the development of infection was unaffected by either gender or age. Neurological status at hospital admission and the severity of the hemorrhage were important factors affecting the development of the infection. More than 80% of patients with a poor neurological status and severe hemorrhage developed an infection during their hospital stay. Finally, there was a significant difference in patient outcome at one year between those patients’ who had developed an infection and those who had not (Table 1).
Table 1. Demographic characteristics of the population studied, comparing the presence or absence of infection during hospitalization.
| |
104 Patient set |
|
|
Infection (N=69) |
No infection (N=35) |
P¹ |
Gender |
|
|
0.339 |
Male n (%) |
22 (21.2%) |
9 (8.7%) |
|
Female n (%) |
47 (45.2%) |
26 (25%) |
|
Age (years) |
|
|
0.479 |
Average (± SD) |
52.2 (± 12.4) |
50.4 (± 12.1) |
|
WFNS score |
|
|
≤ 0.001 |
1,2 n (%) |
30 (28.8%) |
28 (26.9%) |
|
3,4,5 n (%) |
39 (37.5%) |
7 (6.7%) |
|
Glasgow Coma Scale |
|
|
≤ 0.001 |
< 9 n (%) |
27 (26%) |
2 (1.9%) |
|
9-12 n (%) |
9 (8.7%) |
2 (1.9%) |
|
≥ 13 n (%) |
33 (31.7%) |
31 (29.8%) |
|
Modified Fisher Score |
|
|
≤ 0.001 |
0,1 n (%) |
2 (1.9%) |
11 (10.6%) |
|
2,3,4 n (%) |
67 (64.4%) |
24 (23.1%) |
|
Treatment |
|
|
0.278 |
Surgery n (%) |
8 (7.7%) |
6 (5.8%) |
|
Embolisation n (%) |
57 (54.8%) |
29 (27.9%) |
|
No treatment n (%) |
- |
4 (3.8%) |
|
GOS 1 year |
|
|
0.007 |
Good n (%) |
31 (29.8%) |
23 (22.1%) |
|
Poor n (%) |
26 (25%) |
4 (3.8%) |
|
Infection prediction according to blood biomarker concentrations and clinical parameters
In order to evaluate how effectively different blood biomarkers at hospital admission could predict which patients would later develop an infection, we measured the concentrations of SAA, CRP, NP, and WBC in the cohort.
As shown in Table 2, SAA, CRP, and WBC were significantly able to discriminate between patients who did and did not develop an infection. NP was the only biomarker unable to make this distinction at hospital admission.
Table 2. SAA, CRP, NP, and WBC median concentrations at hospital admission according to the presence or absence of infection.
104-Patient set |
|
Infection (n = 69) |
No infection (n = 35) |
P |
SAA (µg/mL) |
181.03 (0-837.2) |
46.42 (1.28-509.55) |
≤ 0.001 |
CRP (µg/mL) |
69.97 (0-362.95) |
34.25 (0.77-217.2) |
0.001 |
NP (nM) |
10.1 (6.02-66.25)) |
9.5 (4.81-21.67) |
0.157 |
WBC (million/mm3) |
13.1 (4.54-24.8) |
11.62 (4.3-27.03) |
0.015 |
AUC analyses were also performed on these four biomarkers (SAA, CRP, NP, and WBC) as well as on different clinical parameters (GCS, WFNS, Fisher, and age) in order to evaluate their accuracy in discriminating between patients with or without infection development.
SAA was the biomarker with the highest accuracy to differentiate between the groups of patients who did and did not develop an infection, reaching a SE value of 26.9% (95% CI, 15.9–38.1) for a SP value of 96.9 (95% CI, 90.6-100). The WFNS was the most effective clinical parameter, with SP values of 97.1 (95% CI, 91.4-100) and SE values of 31.9% (95% CI, 20.3-43.5) (Table 3). When the ROC analyses were performed on these molecules and clinical parameters to calculate the best combination of SP and SE, same results were found; SAA and WFNS were the two most accurate parameters (Supplementary table1).
Table 3. Results of the ROC analysis assessing the capacity of different biomarkers and clinical parameters to differentiate between patients who did and did not develop an infection. The partial AUC, threshold, and SE correspond to SP set at 90%–100%.
Analytes |
SAA (µg/mL) |
2.55 (1.44-5.29) |
1021.42 |
96.9 (90.6 - 100) |
26.56 (15.63 - 37.5) |
CRP (µg/mL) |
1.81 (0.79-4.17) |
176.78 |
96.9 (90.6 - 100) |
19.05 (9.52 - 28.57) |
NP (nM) |
1.06 (0.23-2.73) |
14.59 |
97.14 (88.6 - 100) |
10.14 (4.35 - 17.39) |
WBC (million/mm3) |
0.76 (0-4.61) |
Inf |
100 (100 - 100) |
0 |
Clinical Parameters |
GCS |
2.95 (0.79-5.46) |
6.5 |
97.14 (91.43 - 100) |
31.88 (20.29 - 43.48) |
WFNS |
3.38 (1.62-5.6) |
4.5 |
97.14 (91.43 - 100) |
31.88 (21.74 - 43.48) |
Fisher |
2.46 (1.67-3.79) |
4.5 |
100 (100 - 100) |
14.49 (7.24 - 23.19) |
Age |
0.52 (0.04-2.17) |
80 |
100 (100 - 100) |
1.44 (0 - 4.35) |
To try to improve the accuracy for the prediction of infection, we assessed the possibility of combining all the parameters into panels. In order to select the most promising parameters for inclusion in the panel, we tested all the possible combinations of biomarkers. The iterative permutation-response approach used identified a four-parameter panel including SAA (cut-off: ≥ 427.4 µg/mL), WBC (cut-off: ≥ 13.7), WFNS score (cut-off: >3 and age (cut-off: ≥ 59). An accurate prediction was obtained when at least two of the four parameters were above the cut-off threshold value. The panel exhibited an SE of 64.3% (95% CI, 50–78.6) for an SP value of 100% (95% CI, 100–100) (Figure 1). This results in a significant increase in SE when compared to the best single biomarker (SAA) (p=0.003) and with the best clinical parameter (WFNS) (p=0.004), providing a significant improvement in the capacity to discriminate between patients who will and will not develop an infection.
This prospective study involved 104 patients from two different cohorts. To the best of our knowledge, it is the first to have shown that, for aSAH patients, a predictive test panel comprising clinical parameters and biomarkers measured at hospital admission can significantly improve the prediction of infection compared to the use of single clinical parameters.
2021 Copyright OAT. All rights reserv
The early classification of patients at a higher risk of infection can be essential to improve the management of aSAH patients and their associated outcomes [29,30]. We therefore combined aSAH clinical parameters with known blood diagnostic biomarkers of infection (SAA, CRP, NP, and WBC) in order to find the most effective combination to predict the infection. The combination of SAA, WBC, WFNS, and age showed 100% SP (95% CI, 100–100) and 64.3% SE (95% CI, 50–78.6); interestingly all four parameters included in this panel had already been described individually as markers of infection [12,29,31,32].
The WFNS score, for instance, has been previously proposed as the most significant risk factor and predictor of infection in aSAH patients [1,31,33]. In line with these results, we showed that patients with high WFNS scores were more susceptible to developing an infection than patients with good neurological status (p ≤ 0.001). This could be explained by the fact that ICU patients usually requiring more invasive procedures (mechanical ventilation, urinary and venous catheterization), are more likely to develop nosocomial infections. More importantly, these patients with high values of WFNS present a long suppression of the cellular immune response and an impaired pro-inflammatory response that increases the susceptibility of infection development [34].
It has also been suggested that patients’ age correlates with the presence of pneumonia [5,17,29]. In the present cohort, all the different types of infection (pneumonia, UTI) were evaluated together, and it was not possible to determine the specific impact of pneumonia infections. Nevertheless, the mean age of patients’ developing a nosocomial infection was higher than the age of those who did not, suggesting a positive trend toward the development of infections.
In addition to these two risk factors, the WBC count was also considered in panel generation. In aSAH patients the number of leukocytes have been correlated with the volume of the hemorrhage; being the patients with higher lesion and with higher levels of WBC, the patients with more complications and consequently with poorer outcome [32,35]. In the present study we found that, at hospital admission, patients who developed an infection had significantly higher levels of WBC than patients who did not. This reinforces the hypothesis that patients with poor neurological state at admission are susceptible to develop an infection.
The last parameter included in the panel was SAA—an acute phase inflammatory protein. This blood biomarker is of increasing interest due to its usefulness in monitoring and diagnosing a number of infections [36]. In neutropenic patients with acute leukemia, for example, SAA enabled a differentiation between infectious and non-infectious febrile episodes [37]. In children with a variety of viral infections, SAA concentrations increased during the acute phase of infection [38]. In a small number of aSAH patients it has also been already proposed as an infection marker [12]. In agreement with this, in the present study we have found that, at admission, SAA concentrations in patients who develop an infection were significantly higher than those who did not.
CRP and NP were the other two markers proposed as interesting infection predictors. Nevertheless, their accuracy to distinguish between the two groups of patients was not promising enough to be included in the panel.
CRP is close to SAA in terms of chemical properties; thus, their concentration levels usually follow the same trend across infectious diseases [39-41]. Accordingly, the present study found that, at hospital admission, CRP concentrations in patients developing an infection were significantly higher than in those who did not. Nevertheless, SAA was more accurate thus a more promising biomarker.
On the other hand, NP - a metabolite produced by monocytes/macrophages [42,43] - was initially described as an prognostic outcome biomarker for aSAH [44]. In a previous study, we showed also that NP correlates with the progression of infection from three days after hospitalization [14]. Nevertheless, present results showed that this discrimination capacity was not effective at the admission to the hospital, making from SAA an earlier and most accurate infection marker than NP.
This study showed that combining measurements of SAA, WBC, WFNS, and age significantly improved accuracy over the use of single markers or clinical parameters for the prediction of infection development in aSAH patients. Using the proposed test panel at least six out of ten patients who will develop an infection around five days following an aSAH event could be detected at hospital admission. The potential biochemical pathways occurring after a hemorrhagic event (leukocyte activation, coagulation systems, and complement systems, among others) are too variable and complex to be reflected by any one marker or clinical parameter. Therefore, the combination of biomarkers is more likely to correctly reflect this complex condition.
The panel here described has the potential to be translated, in the future, into an easy to use and interpret clinical tool. However, there are several issues which must be addressed before this important step.
Firstly, our results should be validated on other multicenter cohorts including a larger number of patients to obtain a robust confirmation of the data here presented.
The panel cut-off concentrations here established should be applied in a prospective study in order to confirm the accuracy of the test. Finally, in an effort to reduce the number of episodes of bacterial resistance, antibiotherapy should be reassessed daily; levels of biomarkers below which an infection is considered to be finished should be determined at the different times of hospitalization.
If all of these limitations can be properly addressed, then the prognostic panel presented here could be introduced into clinical practice as a promising tool to improve patient outcomes.
This two-center study showed that the combination of SAA, WBC, WFNS, and age at hospital admission significantly improved the accuracy of single parameters for the prediction of the infection development in aSAH patients. These promising results could ameliorate the management of patients presenting with aSAH in ICUs and their associated outcomes.
The authors declare no financial or other conflicts of interest related to this publication
The authors thank Catherine Fouda and Nadia Walter, as well as the nurses at the Pitié-Salpêtrière Hospital and Charité Universitatsmedizin Hospital for their remarkable technical work and sample collection. Sample collection was funded by the Clinical Research Unit of Paris’ public hospital network. This work was also supported by the Pierre Mercier Foundation for Science.
View supplementary data
- Frontera JA, Fernandez A, Schmidt JM, Claassen J, Wartenberg KE, et al. (2008) Impact of nosocomial infectious complications after subarachnoid hemorrhage. Neurosurgery 62: 80-87. [Crossref]
- Hilker R, Poetter C, Findeisen N, Sobesky J, Jacobs A, et al. (2003) Nosocomial Pneumonia After Acute Stroke: Implications for Neurological Intensive Care Medicine. Stroke 34: 975-981. [Crossref]
- Citerio G, Gaini SM, Tomei G, Stocchetti N, (2007) Management of 350 aneurysmal subarachnoid hemorrhages in 22 Italian neurosurgical centers. Intensive Care Med 33: 1580-1586. [Crossref]
- Douds GL, Tadzong B, Agarwal AD, Krishnamurthy S, Lehman EB, et al. (2012) Influence of Fever and Hospital-Acquired Infection on the Incidence of Delayed Neurological Deficit and Poor Outcome after Aneurysmal Subarachnoid Hemorrhage. Neurol Res Int 2012: 6.
- Laban KG, Rinkel GJE, Vergouwen MDI (2015) Nosocomial infections after aneurysmal subarachnoid hemorrhage: time course and causative pathogens. Int J Stroke 10: 763-766. [Crossref]
- Ulm L, Ohlraun S, Harms H, Hoffmann S, Klehmet J, et al. (2013) STRoke Adverse outcome is associated WIth NoSocomial Infections (STRAWINSKI): procalcitonin ultrasensitive-guided antibacterial therapy in severe ischaemic stroke patients - rationale and protocol for a randomized controlled trial. Int J Stroke 8: 598-603. [Crossref]
- Harms H, Prass K, Meisel C, Klehmet J, Rogge W, et al. (2008) Preventive Antibacterial Therapy in Acute Ischemic Stroke: A Randomized Controlled Trial. PloS one 3: e2158. [Crossref]
- Mayeux R (2004) Biomarkers: Potential Uses and Limitations. NeuroRx 1: 182–188.[Crossref]
- Hong CM, Tosun C, Kurland DB, Gerzanich V, Schreibman D, et al. (2014) Biomarkers as outcome predictors in subarachnoid hemorrhage--a systematic review. Biomarkers 19: 95-108. [Crossref]
- Fluri F, Morgenthaler NG, Mueller B, Christ-Crain M, Katan M (2012) Copeptin, procalcitonin and routine inflammatory markers-predictors of infection after stroke. PLoS One 7: e48309. [Crossref]
- Rallidis LS, Vikelis M, Panagiotakos DB, Rizos I, Zolindaki MG, et al. (2006) Inflammatory markers and in-hospital mortality in acute ischaemic stroke. Atherosclerosis 189: 193-197. [Crossref]
- Azurmendi L, Degos V, Tiberti N, Kapandji N, Sanchez P, et al. (2015) Measuring Serum Amyloid A for Infection Prediction in Aneurysmal Subarachnoid Hemorrhage. J Proteome Res 14: 3948-3956. [Crossref]
- Azurmendi L, Degos V, Tiberti N, Kapandji N, Sanchez P, et al. (2015) Neopterin plasma levels correlate with infection and long-term outcome in aneurysmal subarachnoid haemorrhage. J Neurosurg 124:1287-1299. [Crossref]
- Azurmendi L, Degos V, Tiberti N, Kapandji N, Sanchez-Pena P, et al. (2015) Neopterin plasma concentrations in patients with aneurysmal subarachnoid hemorrhage: correlation with infection and long-term outcome. J Neurosurg 124: 1287-1299. [Crossref]
- Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, et al. (2013) PanelomiX: A threshold-based algorithm to create panels of biomarkers. Translational Proteomics 1: 57-64.
- Turck N, Vutskits L, Sanchez-Pena P, Robin X, Hainard A, et al. (2010) A multiparameter panel method for outcome prediction following aneurysmal subarachnoid hemorrhage. Intensive Care Med 36: 107-115. [Crossref]
- Hoffmann S, Malzahn U, Harms H, Koennecke HC, Berger K, et al. (2012) Development of a clinical score (A2DS2) to predict pneumonia in acute ischemic stroke. Stroke 43: 2617-2623. [Crossref]
- Li Y, Song B, Fang H, Gao Y, Zhao L, et al. (2014) External validation of the A2DS2 score to predict stroke-associated pneumonia in a Chinese population: a prospective cohort study. PLoS One 9: e109665. [Crossref]
- Albrich WC, Harbarth S (2015) Pros and cons of using biomarkers versus clinical decisions in start and stop decisions for antibiotics in the critical care setting. Intensive Care Med 41: 1739-1751. [Crossref]
- Teasdale GM, Drake CG, Hunt W, Kassell N, Sano K, et al. (1988) A universal subarachnoid hemorrhage scale: report of a committee of the World Federation of Neurosurgical Societies. J Neurol Neurosurg Psychiatry 51: 1457. [Crossref]
- Teasdale G, Murray G, Parker L, Jennett B (1979) Adding up the Glasgow Coma Score. Acta Neurochir Suppl (Wien) 28: 13-16. [Crossref]
- Claassen J, Bernardini GL, Kreiter K, Bates J, Du YE, et al. (2001) Effect of Cisternal and Ventricular Blood on Risk of Delayed Cerebral Ischemia After Subarachnoid Hemorrhage:: The Fisher Scale Revisited. Stroke 32: 2012-2020. [Crossref]
- Jennett B, Bond M (1975) Assessment of outcome after severe brain damage. Lancet 1: 480-484. [Crossref]
- Calandra T, Cohen J (2005) The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Crit Care Med 33: 1538-1548. [Crossref]
- van Bussel BCT, Henry RMA, Schalkwijk CG, Ferreira I, Feskens EJM, et al. (2011) Fish Consumption in Healthy Adults Is Associated with Decreased Circulating Biomarkers of Endothelial Dysfunction and Inflammation during a 6-Year Follow-Up. The Journal of Nutrition 141: 1719-1725. [Crossref]
- Klehmet J, Harms H, Richter M, Prass K, Volk HD, et al. (2009) Stroke-induced immunodepression and post-stroke infections: lessons from the preventive antibacterial therapy in stroke trial. Neuroscience 158: 1184-1193. [Crossref]
- Yang YH, Dudoit S, Luu P, Lin DM, Peng V, et al. (2002) Normalization for cDNA microarray data: a robust composite method addressing single and multiple slide systematic variation. Nucleic Acids Research 30: e15. [Crossref]
- Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, et al. (2011) pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics 12: 77. [Crossref]
- Harms H, Grittner U, Dröge H, Meisel A (2013) Predicting post-stroke pneumonia: the PANTHERIS score. Acta Neurol Scand 128: 178-184. [Crossref]
- Grabska K, Gromadzka G, Czlonkowska A (2011) Infections and ischemic stroke outcome. Neurol Res Int 2011: 691348. [Crossref]
- Savardekar A, Gyurmey T, Agarwal R, Podder S, Mohindra S, et al. (2013) Incidence, risk factors, and outcome of postoperative pneumonia after microsurgical clipping of ruptured intracranial aneurysms. Surg Neurol Int 4: 24. [Crossref]
- Parkinson D, Stephensen S (1984) Leukocytosis and subarachnoid hemorrhage. Surg Neurol 21: 132-134. [Crossref]
- Cinotti R, Dordonnat-Moynard A, Feuillet F, Roquilly A, Rondeau N, et al. (2014) Risk factors and pathogens involved in early ventilator-acquired pneumonia in patients with severe subarachnoid hemorrhage. Eur J Clin Microbiol Infect Dis 33: 823-830. [Crossref]
- Sarrafzadeh A, Schlenk F, Meisel A, Dreier J, Vajkoczy P, et al. (2011) Immunodepression after aneurysmal subarachnoid hemorrhage. Stroke 42: 53-58. [Crossref]
- Maiuri F, Gallicchio B, Donati P, Carandente M (1987) The blood leukocyte count and its prognostic significance in subarachnoid hemorrhage. J Neurosurg Sci 31: 45-48. [Crossref]
- Urieli-Shoval S, Linke RP, Matzner Y (2000) Expression and function of serum amyloid A, a major acute-phase protein, in normal and disease states. Curr Opin Hematol 7: 64-69. [Crossref]
- Casl MT, Rogina B, Glojnaric-Spasic I, Minigo H, Planinc-Peraica A, et al. (1994) The differential diagnostic capacity of serum amyloid A protein between infectious and non-infectious febrile episodes of neutropenic patients with acute leukemia. Leuk Res 18: 665-670
- Miwata H1, Yamada T, Okada M, Kudo T, Kimura H, et al. (1993) Serum amyloid A protein in acute viral infections. Arch Dis Child 68: 210-214. [Crossref]
- Nakayama T, Sonoda S, Urano T, Yamada T, Okada M (1993) Monitoring both serum amyloid protein A and C-reactive protein as inflammatory markers in infectious diseases. Clinical Chemistry 39: 293-297. [Crossref]
- Lannergard A, Larsson A, Kragsbjerg P, Friman G (2003) Correlations between serum amyloid A protein and C-reactive protein in infectious diseases. Scand J Clin Lab Invest 63: 267-272. [Crossref]
- Du Clos TW (2000) Function of C-reactive protein. Ann Med 32: 274-278. [Crossref]
- Berdowska A, Zwirska-Korczala K (2001) Neopterin measurement in clinical diagnosis. J Clin Pharm Ther 26: 319-329. [Crossref]
- Murr C, Widner B, Wirleitner B, Fuchs D (2002) Neopterin as a marker for immune system activation. Curr Drug Metab 3: 175-187. [Crossref]
- Mathiesen T, Fuchs D, Wachter H, von Holst H (1990) Increased CSF neopterin levels in subarachnoid hemorrhage. J Neurosurg 73: 69-71. [Crossref]

