Multi-walled carbon nanotubes MWNTs were oxidized by using acid and base Piranha reagent. The acid reagent consists of mixture solution 3:1 H2SO4:H2O2, while the base reagent was NH4OH:H2O2. We will discuss our results by using XRD diffraction also study the effect of oxidizing agents on the surface by measuring the surface area. The ratios of carboxylic groups were discus by making back titration with 0.5 N HCl. The results show a shift in the main of XRD peaks in addition to increasing the agglomerates which causing reduces in the surface area. The effect of oxidizing agents was testing adsorption 40 ppm of Cobalamin which shows enhance with oxidized MWNTs as compare with pristine MWNTs. In addition to MWNTs which treated with acid piranha was more activity to adsorbed Cobalamin than base piranha.
Key word : carbon nanotubes, piranha reagent, xrd diffrection, adsorption
Since Iijima published [1] the first paper of carbon nanotubes CNTs huge amount of attentions were focussed in nanomaterial. The interesting in this nanomaterial related to many reasons, one of them represent by physiochemical properties of carbon nanotubes CNTs, such as mechanism [2], thermal [3], electrical [4], and optoelectronic properties [5]. The other reasons related to many of various applications such as technologies of water purification [6], synthesis of composites [7], hydrogen storage media [8], hydrogen production [9]. The carbon nanotubes are widely used in inorganic chemistry when coordination many legends in complex compound such as Vaska’s compound [10] with metals. The nanotube can be considered as a legend and function with respect to the central metal atom, in coordination complex. Mostly CNTs treated with different techniques and materials before use it which related to abilities the surface of CNTs Firmly resist different Redox reagents which prevent Association with the various components. The oxidation process commonly used for purification [9] and activation the CNTs [11]. The treatments of CNTs either by wet chemical methods [12], photo-oxidation [13], or gas phase treatment [14] has commonly used to purify and activation of the graphitic network. The Piranha mixture [15] one of the reagents that used for this purpose, which done in acidic solution [acidic piranha (H2SO4/H2O2)], or basic solution [basic piranha (NH4OH/H2O2)]. A typical piranha mixture is 3 parts of acid or base with 1 part of hydrogen peroxide solution; other methods may use different ratios of them. Ziegler et al. [16] reported that piranha can be used to cut the tubes of SWNTs under specific and controlled conditions. Datsyuk et al. [17] published comparative studies for oxidant the MWNTs with (H2SO4/H2O2) and (NH4OH/H2O2) as well as many oxidant materials. The (H2SO4/H2O2) showed much lower oxygen content compared to nitric acid oxidation and the absence of added defects on the graphite surface, while (NH4OH/H2O2 complete removal of disordered carbon of the MWNT specimens. Das et al. [18] reported that H2O2 used with different acid and basic compound in purifying and functionalities pristine MWCNTs. Das et al. found that HCl with H2O2 shows more activity than HCl and KOH with H2O2 in the purification process. Kim et al. [19] have treated MWNTs with an NH4OH/H2O2 mixture and showed that the resulting composite with epoxy as matrix had enhanced electrical conductivity due to the minor damages of the graphitic sidewalks. In our earlier work [20] binary composites TiO2/CNTs were analyzed by Raman spectroscopy and X-ray diffraction the work include using SWNTs and MWNTs with TiO2. The analyzing of binary composite TiO2/CNT deals with the effect of SWNTs and MWNTs on the structure of TiO2 and in the activity of binary materials. In these work multi-walled carbon nanotubes MWNTs treated with the piranha reagent in two conditions acidic and basic solution. The effect of oxidation by reagent were characterized by X-ray analysis and measuring the surface area with measuring the carboxylic groups by titration methods. The H2O2 existed in two solutions as complementary materials for reagent with H2SO4 (acid solution) or NH4OH (basic solution). The activities of oxidation were tested in adsorption of 40 ppm Cobalamin at room temperature and makes comparisons with MWNT without oxidant.
Materials
MWNTs, used in this study were purchased from (Aldrich). According to the product specifications, the compound was fabricated by chemical vapor deposition method. The MWNTs 95% carbon nanotubes with mode diameter 5.5 nm. Hydrogen peroxide (30%) from Barcelona-spain. Sulfuric acid (99%) from Merck, Germany and ammonium hydroxide were supplied from Sigma Aldrich.
Method of oxidation MWNT
15 ml of hydrogen peroxide added very slowly to 45 ml of concentrated H2SO4 then allowed to cool before use. 100 mg of multi-walled carbon nanotubes added into 60 ml H2SO4/H2O2 mixture, then stirred for 6 hours at room temperature. The powders were rinsed several times with deionized water and 0.1M NaOH until the pH value reached around 7. After drying, MWNT where heat treatment at 100°C over night by the oven. The product from these steps was referring to it by acid piranha +MWNT. The second sample MWNTs added into NH4OH/H2O2 (45:15) mixture to use the same condition of oxidation and washing the MWNT with deionized water and 0.1M HCl neutralized the solution around 7 units. After that, complete the washing and drying than thermal treatment for the sample which labeled base piranha +MWNT. The process causing produces many active sites on the surface of MWNTs as shown in Figure1.
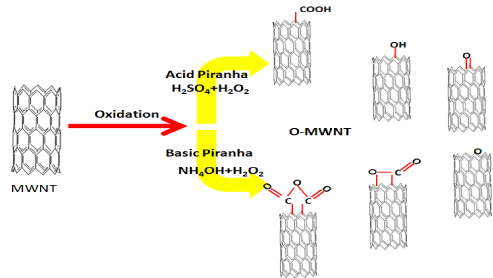
Figure 1. Schematic of the process of reaction MWNTs with acid and base Piranha
XRD analysis
XRD measurements carried out to check the effect of piranha reagent within the structure of MWNTs. The peak at 25.52° represents the characteristic graphitic peak arising due to the presence of the tubular structure of the carbon atoms in the sample with (002) planes (Figure 2). The peak near 43.2° is attributed to the (101) planes of the nanotube structure [21-22]. The oxidized carbon nanotubes by acidic and basic piranha reagent showed two peaks at 14.3° and 16.8° which related to remaining salts which produces from acid and base solution for washing [23]. From Figure 3 the first two peaks before 20° can be related to remaining carbon [24-25] which oxidized by reagent which shows more intensity for MWNTs that oxidized by acid piranha as compare with base piranha.
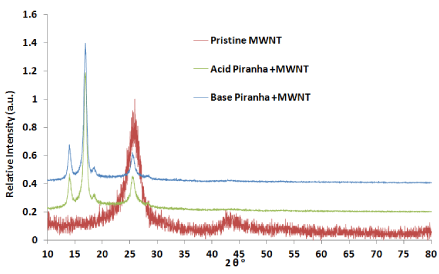
Figure 2.X-Ray diffraction patterns for pristine and oxidized MWNTs by acid and base piranha reagents.
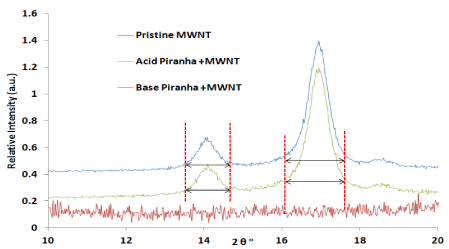
Figure 3.X-ray diffraction patterns for pristine and oxidized MWNTs by acid and base piranha reagents from 2θ =10- 20°.
Figure 4, shows that first main peaks of MWNTs at 25.8° reduced in intensity about four times with acid piranha and five times with base piranha while the shift of the peaks reach to 0.5°. This behavior could be related to increase the particle size of oxidized MWNTs by agglomerates the groups of MWNTs which can calculate by Scherer equation (Figures 5).
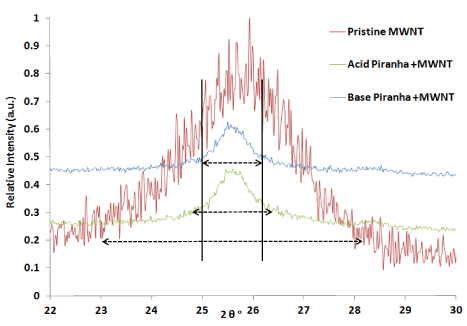
Figure 4.X-Ray diffraction patterns to compare the effect of oxidant reagents for 2θ=22- 30°
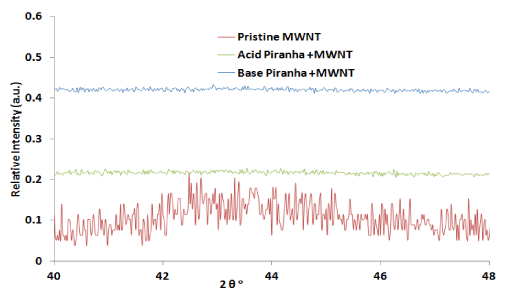
Figure 5.X-ray diffraction patterns for pristine and oxidized MWNTs from 2θ =40° to 2θ =48°
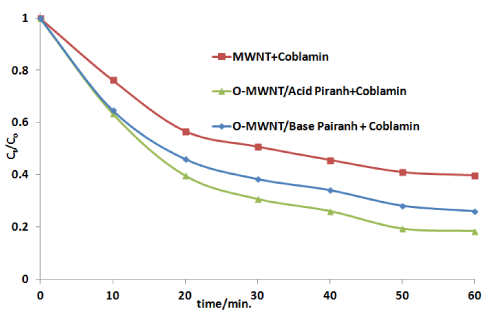
Figure 6.The values of Ct/Co for the adsorption of 40 ppm of Cobalamin by 50 mg/100mL of pristine and oxidized MWNTs by acid and base piranha at 298.15 K
The second main peaks at 43.3° completely removed when oxidized by piranha reagents which may relate to increase the interference between tubular structures when produce oxidation groups. These phenomena enhance the aggregates of MWNTs causing increase the size of groups
X-ray diffraction patterns were used to determine the crystalline size (d) estimated by line broadening measurements in the Debye–Scherrer equation [26]:
d = K λ/β cos θ ……………(1)
Where λ is the X-ray wavelength in nanometers (nm) equal to (0.15405 nm), β is the peak width of the diffraction peak profile at half the maximum height FWHM resulting from small crystallite size in radians, and K is a constant related to the crystallite shape mostly equal to 0.9.
Titration analysis
The titration analysis was done as explained by Datsyuk et. al. [27] when determine the ratios of carboxylic groups. The method depends on measuring the amount (m mol) of carboxylic group on the surface of gram MWNTs. Briefly, the pristine and oxidized MWNTs (50 mg) were dispersed in 50 ml of 0.05N sodium hydroxide with string for 24 h to complete the equilibrium between carboxylic and hydroxyl groups. After that, non-reactant sodium hydroxide was calculated by back titration with 0.05 N of hydrochloric acid. The results show that effect of acid piranha reagent to create carboxylic groups were more than basic piranha and pristine MWNTs when shows 1.79 m mol.g-1, 1.5 m mol.g-1 and 1.4 m mol.g-1 respectively.
From reported results in Table 1, the particle size of oxidized MWNTs increase to more than double values as compared with pristine MWNTs. Increase the practical size effect on the value of surface area for the materials after treatment with oxidized reagent, which was more for base piranha than acid piranha. At the same time the results of titration show that small value of carboxylic groups with MWNTs which oxidized with base piranha. The reduce in carboxylic groups related to creating a base group on the surface of MWNTs which enhance the agglomerations and reduces the surface area.
Table 1. The value of FWHM for characterized peak (2θ≈25°) and the particle size with surface area for pristine and oxidized MWNTs
| Sample |
2 θ° |
FWHM |
D002 |
SBET (m2/g) |
| Pristine MWNT |
25.52 |
2.2630 |
3.58 |
282 |
| Acid Piranha+ MWNT |
25.65 |
0.8494 |
9.55 |
175 |
| Base Piranha+ MWNT |
25.63 |
0.8033 |
10.09 |
146 |
Measuring the activity
The activity of the new changing surface was tested by adsorption [21] Cobalamin (C63H88CoN14P), which is vitamin B12, as a complex compound of an organometallic species with a cobalt atom in the Corrine ring [28]. The typical adsorption experiments of 40 ppm Cobalamin molecules with 0.05 g of pristine and oxidized MWNTs in aqueous phases. The pH equal to 7.8 at 298.15K by adding 1N NaOH or HCl, with using magnetic mixer at 475 rpm. The required amount of the adsorbent where suspend in100 cm3 of an aqueous solution of Cobalamin; in various times 2 ml taken from the reaction vessel, and filtered to remove the particles by centrifuge at 4,000 rpm for 10 minutes in an 800 B centrifuge. The Cobalamin absorbance was measured at 550 nm, using a Shimadzu Cary 100Bio UV – visible spectrophotometer.
Table 2. The value of rate adsorption and compare for the synergy factor for adsorption process of Cobalamin by pristine e and oxidized MWNTs.
| Sample |
K(s-1) |
R |
| Pristine MWNT |
0.0183 |
1 |
| Acid Piranha+ MWNT |
0.0259 |
1.41 |
| Base Piranha+ MWNT |
0.0331 |
1.80 |
The adsorption experiments of the Cobalamin solutions follow the pseudo first-order kinetics with respect to the concentration of dyestuff in the bulk solution (C0):
ln(C0/Ct)=k1*t …………….(2)
Where C0 refer to the initial concentration in the bulk solution before starting the adsorption process, C refers to concentration of Cobalamin after t min of adsorption. The k1 is the adsorption rate constant. A plot of ln(C0/C) versus t for Cobalamin adsorption with pristine and oxidized MWNTs with acid and basic reagent is presented in Figure7. The value of k1 can be obtained directly from the slope of the respective linear curves in the plot which listed in Table 1
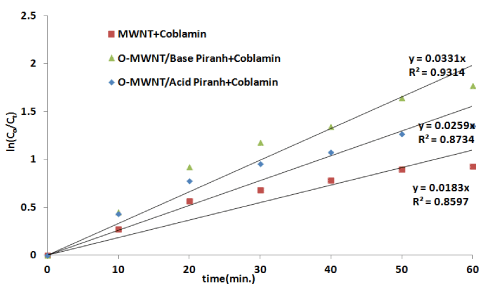
Figure 7.Rate constant for pristine and oxidized (0.05 g) MWNTs with 40 ppm Cobalamin at 298.15 K
The synergy factor ( R ) [29] is defined as the following equations:
R = k (O-MWNT)/ k (pristine-MWNT) …………(3)
When k (O-MWNT), k (pristine-MWNT) refer to the adsorption rate constant for remove Cobalamin in exist of oxidized and pristine MWNTs respectively.
The results showed that MWNTs after the oxidation succeed in adsorption the Cobalamin
O-MWNT (acid piranha) ˃ O-MWNT(base piranha)˃ MWNT
Chemical oxidations of MWNTs with acid and base piranha reagents were studied. The reference sample was pristine Aldrich MWNTs which contains very low amount of graphite nanoparticles without any functionalized oxygen groups. The oxidative reagents used were acidic piranha solution (H2SO4/H2O2) and basic piranha solution (NH4OH/H2O2). After each treatment, the functionalization yield, namely the density of carboxyl and hydroxyl functional groups in the graphitic network, was quantified by titration measurements. The results show that acidic piranha was succeeding in the creation of many groups of carboxylic acid, which was more than basic piranha and pristine MWNTs. The effect of oxidation enhances the adsorption of Cobalamin by creating more functional groups on the surface and reduce the agglomeration. For that XRD analysis, which refers to agglomeration and ratios of functional groups by titration explained preference the activity of acidic piranha as compare with base and pristine MWNTs.
- Iijima S (1991) Helical microtubules of graphitic carbon. Nature 354: 56–58.
- Vigolo B, Pénicaud A, Coulon C, Sauder C, Pailler R, et al. (2000) Macroscopic fibers and ribbons of oriented carbon nanotubes. Science290: 1331-1334. [Crossref]
- Itkis ME, Borondics F, Yu A, Haddon RC (2007) Thermal conductivity measurements of semitransparent single-walled carbon nanotube films by a bolometric technique. Nano Lett 7: 900-904. [Crossref]
- Liu J, Rinzler AG, Dai H, Hafner JH, Bradley RK, et al. (1998) Fullerene pipes. Science280: 1253-1256.
- Whitby RLD, Fukuda T, Maekawa T, James SL, Mikhalovsky SV(2008) Geometric control and tuneable pore size distribution of buckypaper and buckydiscs. Carbon 46: 949–956.
- Ali ME, Das R, Maamor A, Hamid SBA (2014) Multifunctional carbon nanotubes (CNTs): a new dimension in environmental remediation. Advanced Materials Research 832: 328–332.
- Parveen S, Rana, R Fangueiro( 2013) A review on nanomaterial dispersion, microstructure, and mechanical properties of carbon nanotube and nanofiber reinforced cementitious composites. Journal of Nanomaterials.
- Sahaym U, Norton MG (2008) Advances in the application of nanotechnology in enabling a hydrogen economy. Journal of Materials Science 16: 5395–5429.
- Khan G, Kim YK, Choi SK, Han DS, Abdel-Wahab A, et al. (2013) Evaluating the catalytic effects of carbon materials on the photocatalytic reduction and oxidation reactions of TiO2. BullKorean ChemSoc.
- Sarbajit Banerjee and Stanislaus S Wong (2002) Functionalization of carbon nanotubes with a metal-containing molecular complex. American Chemical Society 2: 49-53.
- Feng Y, Zhang H, Hou Y, McNicholas TP, Yuan D, et al. (2008) Room temperature purification of few-walled carbon nanotubes with high yield. ACS Nano 2: 1634-1638. [Crossref]
- Yu WX, Sheng JY , Hong Z, Chang ZJ 2021 Copyright OAT. All rights reservodified multi-walled carbon nanotubes supported Pt-Mo electrocatalyst. ChemRer Chinese Universities27: 486-489.
- Das R, Eaqub Ali Md, Bee Abd Hamid S, Annuar MSM, Ramakrishna S (2014) Common wet chemical agents for purifying multiwalled carbon nanotubes. Journal of Nanomaterials.
- Grujicic M, Gao G, Rao AM, Tritt TM, Nayak S (2003) UV-light enhanced oxidation of carbon nanotubes. Appl Surf Sci 214: 289–303.
- Tsang SC, Harris PJ, Green ML (1993) Thinning and opening of carbon nanotubes by oxidation using carbon dioxide. Nature 362: 520–522.
- Yuin Chia JS, Michelle TT, Tan JK, Chin PS, Khiew, et al. (2012) A Parametric study on the synthesis of graphene using Piranha-like thermal exfoliation, IPCBEE.
- Ziegler KJ1, Gu Z, Peng H, Flor EL, Hauge RH, et al. (2005) Controlled oxidative cutting of single-walled carbon nanotubes. J Am Chem Soc 127: 1541-1547. [Crossref]
- Datsyuk V, Kalyva M, Papagelis K, Parthenios J, Tasis D, et al. (2008) Chemical oxidation of multiwalled carbon nanotubes. Carbon46: 833–840.
- Rasel Das, Md. Eaqub Ali, Sharifah Bee Abd Hamid, MSM Annuar, et al. (2014) Common wet chemical agents for purifying multiwalled carbon nanotubes, hindawi publishing corporation Journal of Nanomaterials.
- Kim YJ, Shin TS, Choi HD, Kwon JH, Chung YC, et al. (2005) Electrical conductivity of chemically modified multiwalled carbon nanotube/epoxy composites. Carbon 43: 23–30.
- Firas H, Abdulrazzak, Falah H Hussein (2015) Effects of nanoparticle size on catalytic and photocatalytic activity of carbon nanotubes-titanium dioxide composites. J Environ Anal Chem 2: 2.
- Nishimura K, Okazaki N, Pan L, Nakayama Y (2004) In situ study of iron catalysts for carbon nanotube growth using X-ray diffraction analysis. Japanese Journal of Applied Physics 43: 471–474.
- Capula S, Aguir K, Cervantes F, Villa L, Moncayo JA, et al. (2014) Ozone sensing based on palladium decorated carbon nanotubes. Sensors: 6806-6818
- Ashcroft, Neil W, Mermin ND (1976) Solid State Physics. New York: Holt, Rinehart and Winston.
- Manoj B, Kunjomana AG (2012) Study of stacking structure of amorphous carbon by X-ray diffraction technique. Int J ElectrochemSci 7: 3127–3134.
- Leroy A, Harold PK (1950) Determination of crystallite size with the X-ray spectrometer. J AppPhys 21: 126-7.
- Datsyuk V, Kalyva M, Papagelis K, Parthenios J, Tasis D, et al. (2008) Chemical oxidation of multiwalled carbon nanotubes. Carbon46: 833-840.
- Quaroni L, Reglinski J, Smith W (1995) Surface enhanced resonance Raman scattering from cyanocobalamin and 5’-deoxyadenosylcobalamin. J Raman Spect 26: 1075-1076.
- Ying Y, Jimmy C Y, Cho-Yin C, Yan-Ke C, Jin-Cai Z, et al. (2005) Applied catalysis B: Environmental 61 1.







