Abstract
Clinical and preclinical findings have suggested a role for ascorbate in mood disorders. The present study was designed to investigate the effect of acute and chronic ascorbate treatment in animal models of memory, anxiety and depression using discriminative avoidance, forced swimming, light-dark box and openfield tests. Aged male Wistar rats (18 months) were treated with saline or ascorbate (50, 500 or 1000 mg/kg body weight) during 25 days. The acute treatment did not improve learning and memory parameters evaluated in the plus maze discriminative avoidance test. Besides, in the training session, ascorbate (1000 mg/kg) decreased the time spent in the open arms and 500 and 1000 mg/kg doses increased the time spent in the nonaversive enclosed arm, indicating anxiety-like behavior. On the 10th day of treatment, ascorbate (1000 mg/kg) showed an antidepressant-like activity, since the treated animals showed decreased immobility time when compared to the saline group in the forced swimming test. The treatment with ascorbate did not alter any parameters in the light-dark box test evaluated on the 15th day; however, on the 25th day, ascorbate (500 mg/kg and 1000mg/kg) decreased the number of entries into peripheral squares and the number of rearings in the open field test, suggesting an anxiolytic-like profile. In conclusion, our findings suggest that acute ascorbate treatment may induces an anxiety state, while at long term seems to exert anxiolytic activity, as well as exhibiting an antidepressant-like activity in aged rats.
Key words
Antidepressant-like effect, anxiety, ascorbate, aged rats
Introduction
Depression and anxiety disorders are high-prevalence mental health conditions with negative psychosocial impact. The co-occurrence of depression and anxiety disorders is extremely frequent in primary care [1]. Considering that major depressive disorder is an illness of longer duration as well thatavailable treatments are inadequate for many individuals and induce various side effects [2], different treatment approaches have been sought.
Clinical findings have been suggested that ascorbate might exert an important role in mood disorders. In this context, there is evidence that an inadequate ascorbate intake and subsequent it reduced plasma levels could be linked to the pathophysiology of depression [3]; while a chronic treatment with ascorbate reversed both depression and scurvy states [4]. Indeed, Coochi and colleagues [5] showed that ascorbate treatment for two weeks lead to complete remission from depression symptoms in depressed patients. Furthermore, Brody, et al. [6] also reported that a high dose of ascorbate for 14 days in humans elicited a reduction in the state of anxiety and also decreased scores of depression in healthy young adults, which can be considered as indicative of mood improvement.
It is interesting to note that the brain maintains high and regulated levels of ascorbate [7], where it is proposed to exert neuroprotective effects [8]. In addition, ascorbate is considered to be an important neuromodulator, modulating both dopamine- and glutamate-mediated neurotransmission [7-10]. Also, ascorbate exerts other relevant biological functions, including acting asa cofactor for important hydroxylation reactions needed for the synthesis of catecholamines [11] and playing the role of a water-soluble antioxidant [10].
Although clinical studies suggest an antidepressant activity for ascorbate, there arefewstudies describing the psychopharmacological profile of ascorbate in animal models [12]. It was recently demonstrated an antidepressant-like effect following subchronic ascorbate administration in mice [13]; however, it will be interesting investigate this effect in other rodent models such as Wistar rats.
Several studies also show an age-related decrease in brain ascorbate levels [14]. In fact, we previously showed a remarkable decrease on ascorbate uptake in hippocampi of aged rats [15]. It is interesting to note that there is a strong association between aging and depression, with numerous clinical studies showing that the prevalence of depression increases in old age. In fact, depression is three times as frequent in the elderly as it is in the rest of the population [16-18]. Despite these findings, studies regarding the effect of ascorbate treatment on psychopharmacological profile in aged rats are lack.
Therefore, the present study was designed to investigate the acute and chronic effects of ascorbate treatment in animal models of memory, anxiety and depression in aged Wistar rats.
Material and methods
Animals
Male Wistar rats at 18 months of age were obtained from our breeding colony and maintained under controlled light and environmental conditions (12 h light/12 h dark cycle at 22±2°C), with food and water ad libitum, in Biotério Setorial of the Departamento de Farmacologia, Instituto de Ciências Básicas da Saúde, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS. All manipulations were carried out between 11:00 and 18:00 hours. In all experiments, the animals were observed in a random order, in a blind manner and always by the same experimenter. After each behavioral session, the apparatus was cleaned with 90% alcohol solution. Procedures were performed in accordance with the NIH Guide for the Care and Use of Laboratory Animals and Local Ethics Committee approved all handling and experimental conditions. In addition, all efforts were made to minimize animal suffering and the number of animals needed in this work; we use the same animals in all tests in an attempt tomaximize the data gained from an individual animal in compliance with ethical principles. Different anxiety tests were performed in order to avoid learning (Figure 1).
Figure 1. Schematic timeline showing the behavioral tests performed in this study
Treatment
The ratswere randomly assigned to four experimental groups: control,and ascorbate at different concentrations: 50, 500 or 1000 mg/kg body weight (for each dose8-12/group).They were treated once a day, with saline or ascorbate by gavage for 25 days. All tests were performed in a quiet and darkened room.
Plus-maze Discriminative Avoidance Task
The plus-maze discriminative avoidance task was performed as previously described, concomitantly evaluating memory and anxiety-like behavior [19,20]. The apparatus employed was a modified elevated plus-maze, made of acrylic, containing two enclosed arms with side walls and no top (50x50x10 cm), opposite two open arms (50x10 cm). A 100-watt lamp was placed exactly over the middle of one of the enclosed arms (aversive enclosed arm). The temperature in all arms of the apparatus was the same as that of the experimental room (22–23°C). The test was performed in 2 days, as follows. First, in the training session (day 1), each rat was placed in the center of the apparatus and, over a period of 10 minutes, every time the animal entered the enclosed arm containing the lamp, an aversive situation was produced and maintained until the animal left the arm. The aversive stimuli were the 100-watt light and an 80-dB noise produced by a small machine placed under the aversive enclosed arm; these were turned on (by the experimenter) only when the rat entered the aversive enclosed arm. In the test session (day 2), the rats were again placed in the apparatus for 3 minutes, without receiving the aversive stimulation. Rats received saline or ascorbate and 5 min later were submitted to the apparatus, both in training (day 1) and test (day 2) sessions [19]. Learning and memory was evaluated by the time spent in the aversive enclosed arm in the test session. The anxiety-like behavior was evaluated by the total number of entries and time spent in any of the arms, total number of head dipping, rearing, and grooming and fecal boli.
Forced Swimming Test
The procedure used was that of Porsolt, et al. [21] modified by Detke and Lucki [22]. On the 10th day, the rats were placed in individual Plexiglas cylinders (diameter: 34 cm, height: 30 cm), that had previously been filled with water (23–25 °C) to a depth of 20 cm. The procedure consisted of two swimming sessions: first, a 15-minute pre-test, which was followed 24 hours later by a 5-minute test. Rats were considered as immobile when floating motionless or making only those movements necessary to keep the head above water. Following the training and the test session, the animals were dried and returned to their cages. The water was changed and the cylinder rinsed with clean water after each rat. A decrease in the duration of immobility during the forced swimming test was taken as a measure of antidepressant activity.
Light-Dark Box Test
The Light-Dark Box was as described previously by Chaouloff, et al. [23]. The apparatus consisted of two acrylic compartments, one dark (15.5x13.5 cm) and one white (15.5x17.5 cm). The floor was divided into squares, and an interconnecting tunnel (7x10 cm) allowed the rat to go from one compartment to the other. The lateral walls and floor of the light compartment were white, and a single, 60-W lamp bulb provided illumination from above. The dark compartment was black. The apparatus was positioned on a bench 70 cm above the floor. On the 15th day, each rat was tested by placing it in the center of the white compartment, facing away from the dark compartment, and then being allowed to explore the novel environment for 5 minutes. The number of head dipping, rearing, and grooming and fecal boli, time spent in each compartment, number of squares, number of transitions between the compartments and the latency of the initial movement to the dark compartment. A rat was considered to have entered the new compartment when all four legs were in this area [24].
Open Field Test
The Open-Field apparatus was based on that described by Walsh and Cummins [25]. Briefly, the apparatus consisted of a wooden box measuring 60 ×40 ×50 cm with a glass front wall and a floor that was divided by black lines into 12 equal squares. On the 25th day, the animals were gently placed facing the rear left corner of the arena and the behaviors recorded during the 5 minutes of the test session were: the number of squares crossed, time spent in peripheral and central squares, and the number of rearings, grooming and fecal boli.
Statistical analysis
The results were expressed as median (25th/75th percentiles) or mean (± S.E.M.) values. Before statistical testing data were explored to assess their pattern of distribution. The Kruskal-Wallis or One-Way Analysis of Variance (ANOVA), followed by Dunn or Tukey Tests, respectively, was employed considering data distribution. Differences were considered as significant at p < 0.05.
Results
The acute treatment with different doses of ascorbate did not improve learning and memory parameters evaluated in the plus maze discriminative avoidance task.In the training session, all groups spent more time in the nonaversive enclosed arm than in the aversive enclosed arm (F(1,76)=45.564; p=0.007). All groups spent a similar time in both enclosed arms in the test session; the treatment with ascorbate did not alter the percent time in the aversive arms in either session (data not shown).
In addition, in the training sessionfor the plus maze discriminative avoidance task the effects of ascorbate on anxiety-related behavior (open-arm avoidance) were observed. Treatment with ascorbate (1000 mg/kg) decreased the time spent in the open arms and 500 and 1000 mg/kg doses increased the time spent in the nonaversive enclosed arm (respectively, p=0.04 and p<0.001, Figure 2), indicating an increase in anxiety-like behavior. The treatment with ascorbate (500 and 1000 mg/kg) had no effect on locomotor activity in this task, expressed as the total number of entries in both arms (Table 1).
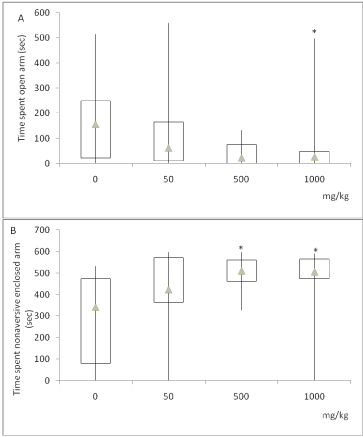
Figure 2. Effects of acute ascorbate treatment on rats tested in the plus maze discriminative avoidance task. Time spent in open arms (Panel A) and in the nonaversive enclosed arms (Panel B). *Values significantly different from control group (0 mg/kg), p<0.05 (Kruskal-Wallis followed by Dunn)
On the 10th day of treatment, ANOVA indicated a significant difference among treatments (F(3.36) = 4.179 p=0.013), the highest dose of ascorbate (1000 mg/kg) decreased the immobility time when compared to the saline group (Figure 3). This result indicated an antidepressant-like activity of ascorbate. The treatment with ascorbate did not alter any parameters in the light-dark box test, another anxiety model, evaluated on the 15th day.
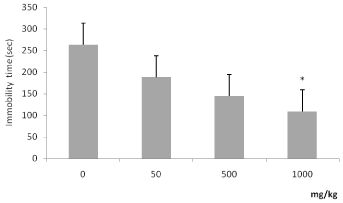
Figure 3. Effects of chronic ascorbate treatment on immobility time in rats tested in the
forced swimming test. ANOVA followed by Tukey. *Value significantly different from control group (0 mg/kg), p<0.05
The data from the open field test are shown in Figure 4. On the 25th day, ascorbate groups (500 mg/kg and 1000mg/kg) showed a decrease in the number of entries into peripheral squares when compared with control (Figure 4A, ANOVA followed by Tukey’s; F(3,44) = 4.252; p=0.011). The treatment with ascorbate decreased the number of rearings when compared with control (Figure 4B, Kruskal-Wallis followed by Dunn’s; H=14.94; p=0.002). These findings indicate that the chronic ascorbate treatment exhibited an anxiolytic-like profile in the open field test.
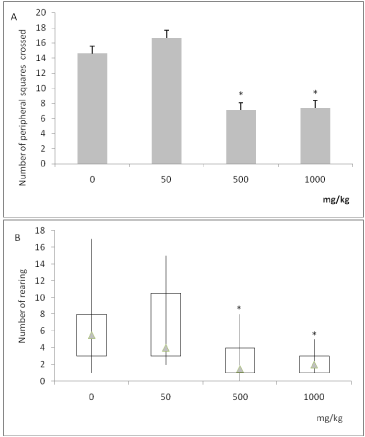
Figure 4. Effects of chronic ascorbate treatment on rats exposed to the open-field test. Number of entries into peripheral squares (Panel A, ANOVA followed by Tukey). Number of rearings (Panel B, Kruskal-Wallis followed by Dunn). *Values significantly different from control group (0 mg/kg), p<0.05
Discussion
Our data corroborate findings that have suggested a role for ascorbate in mood disorders [3-6,12]. Ascorbate administration induced both anxiety and anxiolytics states, respectively, in the short- and long-term, as well as producing an antidepressant-like activity in aged rats.
Ascorbate is an important water-soluble antioxidant, which scavenges oxygen or nitrogen-centered radical species generated during normal metabolism, such as peroxyl radicals, superoxide, singlet oxygen, peroxynitrite and hydroxyl radicals. Several lines of evidence indicate that ascorbate is localized preferentially in neurons [7]. Considering that a long-term dietary supplementation with various antioxidants has been found to retard the onset of age-related deficits in neural functioning [26-28], the present study examined learning and memory behavior. Acute ascorbate administration did not alter any learning and memory parameters (aversive arms) in the discriminative avoidance test. Similarly, ascorbate did not improve memory when administered alone in aged (15 months) and young adult mice (3 months); however, it was effective when given in association with vitamin E [29]. On the other hand, the acute ascorbate-induced anxiety state described here may be a confounding factor in memory and learning evaluation and it seems possible that chronically ascorbate could be a modulator of learning and memory.
The acute administration of ascorbate produces an anxiogenic-like effect in the plus maze discriminative avoidance task; since ascorbate decreased the time spent in the open arms and increased the time spent in the nonaversive enclosed arm. This result cannot be explained by changes in locomotor activity, since acute administration of ascorbate did not modify locomotion in this task.
Although the selective serotonin reuptake inhibitors have been reported to be effective treatments for generalized anxiety disorder, previous studies have demonstrated that acute treatment with antidepressants induces anxiogenic-like effects [30,31]. Hascoët and collaborators [32] showed that two specific inhibitors of norepinephrine reuptake, desipramine and maprotiline, and the atypical antidepressant trazodone demonstrated anxiogenic-like effects in the four-plate test, an animal model of anxiety based on stress caused by unconditioned fear. Furthermore, acute administration of fluoxetine, a selective serotonin reuptake inhibitor, induces an anxiety state in a variety of behavioral tests such as immobilization stress and the social interaction test [33].
These findings corroborate clinical observations showing that selective serotonin reuptake and selective serotonin and norepinephrine reuptake inhibitors cause anxiety states in the early stages of treatment [34]. Indeed, a single dose ofcitalopram has been found to increase both the alarm reaction [35] and anxiety in healthy volunteers [36]. This effect of antidepressants may be associated with a serotonergic mechanism, for example selective blockade of the reuptake transporter, resulting in an increase in 5HT concentrations [37]. Moreover, the 5HT2A/2C, 5HT3 and 5HT1A receptor subtypes are especially relevant to this discussion, given their implication in both anxiety and depression [38,39]. Acute anxiogenic-like effects of fluoxetine, sertraline, citalopram or m-CPP, a nonselective 5-HT2C receptor agonist, were reversed by administration of the 5-HT2C receptor subtype selective antagonist, suggesting that the activation of 5-HT2C receptors might mediate, at least in part, the increased anxiety as a consequence of increased synaptic serotonin concentrations induced by these drugs [33,40].
The anxiogenic-like effect of ascorbate may be involved with its neuromodulatory function. It has been reported that brain areas that show the highest contents of ascorbate are rich in catecholamine innervations [41]. It is also well established that ascorbate is essential for catecholamine biosynthesis in neural tissues, serving as a cofactor for dopamine β-hydroxylase in the conversion of dopamine to norepinephrine. Furthermore, ascorbate is implicated in the regulation of acetylcholine and catecholamine release from synaptic vesicles [42].
Besides, a systemic effect could be involved, since ascorbate is co-released with catecholamines in the adrenal medulla [9]. Indeed, the adrenal cortex contains a higher concentration of ascorbic acid than other tissues, in both human and animals. We suggest that the acute anxiogenic-like effect of ascorbate may be involved with an increase on catecholamine levels, considering its role in anxiety states. These biochemical properties might be related to inhibition of Na+, K+- ATPase activity induced by ascorbate, as well as inhibition of voltage-dependent K1 currents through a Gs-protein–PKA system [43-45].
Interestingly, chronic treatment with ascorbate reduced the immobility time in the forced swim test, an animal model of depression. It is relevant to note that this result is consistent with previous studies in mice [12]. The forced swim test has predictive validity [46], being one of the most commonly used methods to detect and characterize the efficacy of tricyclic antidepressants, monoamine oxidase inhibitors, and atypical antidepressants such as mianserin [47]. Selective serotonin and selective noradrenaline re-uptake inhibitors reduced immobility through sub-acute and chronic treatment [48]. The antidepressant-like effect observed with chronic ascorbate in this work might be associated temporally with the therapeutic effects of antidepressant treatment.
It is interesting to note that on the 15th day ascorbate did not alter any parameters in the light-dark box test, which may suggest tolerance to the anxiogenic effect of ascorbate. Griebel and colleagues [49] also demonstrated that repeated administration of mCPP induces complete tolerance to its anxiogenic-like activity in rats in the elevated zero-maze test after both subchronic and chronic administration.
Afterward, chronic ascorbate administration demonstrated an anxiolytic effect in the open field test, given that the higher doses decreased thigmotaxis. Peripheral movement or thigmotaxis, an important anxiety-related behavior observed in this test [50], is sensitive to the action of anxiolytic drugs such as benzodiazepines [51,52], and it is important to note that the validity of thigmotaxis in measuring emotionality has been demonstrated in mice [53,54] as well as in rats [50,55]. In addition to thigmotaxis, several other parameters evaluated in the open field test have also been employed to measure emotionality, such as the number of rearings [56]. In this work the chronic treatment with ascorbate increased the number of rearings, which represents further evidence for its anxiolytic effect, since the treatment with anxiolytic drugs causes an increase on number of rearing [57]. However, ascorbate did not affect locomotor activity of rats in the open field test.
Conclusion
Our findings demonstrated that acute administration with ascorbate induced an anxiety state, while chronically seems to exert an antidepressant and anxiolytic-like activities in aged rats. These data might propose that ascorbate has a similar profile of action to that of antidepressant drugs and could be an alternative approach to depression and anxiety treatment. However, further behavioral tests, neurochemical assays and also clinical studies will be necessary to confirm the antidepressant and anxiolytic effects of ascorbate and to clarify its mechanism of action.
Acknowledgements
This work was supported by the Brazilian funding agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq (Dr. I.R.Siqueira, Viviane Elsner, Felipe Moysés – 2009); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - CAPES (F. Moysés – 2008). Karine Bertoldi was supported by the National Program of Undergraduate Research Studentships (Bolsa de Iniciação Científica, FAPERGS).
References
- Kessle RC, Nelson CB, McGonagle KA, Liu J, Swartz M, et al. (1996) Comorbidity of DSM-III-R major depressive disorder in the general population: results from the US National Comorbidity Survey. Br J Psychiatry Suppl 30: 17-30. [crossref]
- Nestler EJ, Barrot M, DiLeone RJ, Eisch AJ, Gold SJ, et al. (2002) Neurobiology of depression. Neuron 34: 13-25. [crossref]
- Khanzode S., Khanzode SD, Dakhale GN (2003) Serum and plasma concentration of oxidant and antioxidants in patients of Helicobacter pylori gastritis and its correlation with gastric cancer. Cancer Lett 195: 27-31. [crossref]
- Chang CW, Chen MJ, Wang TE, Chang WH, Lin CC, et al. (2007) Scurvy in a patient with depression. Dig Dis Sci 52: 1259-1261. [crossref]
- Cocchi P, Silenzi M, Calabri G, Salvi G (1980) Antidepressant effect of vitamin C. Pediatrics 65: 862-863. [crossref]
- Brody S, Preut R, Schommer K, Schürmeyer TH (2002) A randomized controlled trial of high dose ascorbic acid for reduction of blood pressure, cortisol, and subjective responses to psychological stress. Psychopharmacol 159: 319-324. [crossref]
- Rice ME (2000) Ascorbate regulation and its neuroprotective role in the brain. Trends Neurosci 23: 209-216. [crossref]
- Padayatty SJ, Katz A, Wang Y, Eck P, Kwon O, et al. (2003) Vitamin C as an antioxidant: evaluation of its role in disease prevention. J Am Coll Nutr 22: 18-35. [crossref]
- Grünewald RA1 (1993) Ascorbic acid in the brain. Brain Res Brain Res Rev 18: 123-133. [crossref]
- Rebec GV, Pierce RC (1994) A vitamin as neuromodulator: Ascorbate release into the extracellular fluid of the brain regulates dopaminergic and glutamatergic transmission. Prog Neurobiol 43: 537-565. [crossref]
- Padh H (1990) Cellular functions of ascorbic acid. Biochem Cell Biol 68: 1166-1173. [crossref]
- Binfaré RW, Rosa AO, Lobato KR, Santos AR, Rodrigues AL (2009) Ascorbic acid administration produces an antidepressant-like effect: evidence for the involvement of monoaminergic neurotransmission. Prog Neuropsychopharmacol Biol Psychiatry 33: 530-540. [crossref]
- Moretti M, Budni J, Ribeiro CM, Rieger DK, Leal RB, et al. (2016) Subchronic administration of ascorbic acid elicits antidepressant-like effect and modulates cell survival signaling pathways in mice. J Nutr Biochem 38: 50-56. [crossref]
- Sahoo A, Chainy GB (1997) Alterations in the activities of cerebral antioxidant enzymes of rat are related to aging. Int J Dev Neurosci 15: 939-948. [crossref]
- Siqueira IR, Elsner VR, Leite MC, Vanzella C, Moysés Fdos S, et al. (2011) Ascorbate uptake is decreased in the hippocampus of ageing rats. Neurochem Int 58: 527-532. [crossref]
- Roberts RE, Kaplan GA, Shema SJ, Strawbridge WJ (1997) Does growing old increase the risk for depression? Am J Psychiatry 154: 1384-1390. [crossref]
- Blazer DG (2002) Self-efficacy and depression in late life: a primary prevention proposal. Aging Ment Health 6: 315-324. [crossref]
- Colasanti V, Marianetti M, Micacchi F, Amabile GA, Mina C (2010) Tests for the evaluation of depression in the elderly: a systematic review. Arch Gerontol Geriatr 50: 227-230. [crossref]
- Frussa-Filho R, Kameda SR, Carvalho RC, Takatsu-Coleman AL, Ricardo VP, et al. (2007) Dissociation of the effects of ethanol on memory, anxiety, and motor behavior in mice tested in the plus-maze discriminative avoidance task. Psychopharmacol 192: 39–48. [crossref]
- Kameda SR, Frussa-Filho R, Carvalho RC, Takatsu-Coleman AL, Ricardo VP, et al. (2007) Dissociation of the effects of ethanol on memory, anxiety, and motor behavior in mice tested in the plus-maze discriminative avoidance task. Psychopharmacol 192: 39-48. [crossref]
- Porsolt RD, Anton G, Blavet N, Jalfre M (1978) Behavioural despair in rats: a new model sensitive to antidepressant treatments. Eur J Pharmacol 47: 379–391. [crossref]
- Detke MJ, Lucki I (1996) Detection of serotonergic and noradrenergic antidepressants in the rat forced swimming test: the effects of water depth. Behav Brain Res 73: 43-46. [crossref]
- Chaouloff F, Kuliko2021 Copyright OAT. All rights reserv (1995) Male Fischer 344 and Lewis rats display differences in locomotor reactivity, but not in anxiety-related behaviours: relationships with the hippocampal serotonergic system. Brain Res 693: 169-178. [crossref]
- Andrade TG, Graeff FG (2001) Effect of electrolytic and neurotoxic lesions of the median raphe nucleus on anxiety and stress. Pharmacol Biochem Behav 70: 1-14. [crossref]
- Walsh RN, Cummins RA (1976) The Open-Field Test: a critical review. Psychol Bull 83: 482-504. [crossref]
- Bickford PC, Gould T, Briederick L, Chadman K, Pollock A, et al. (2000) Antioxidant-rich diets improve cerebellar physiology and motor learning in aged rats. Brain Res 866: 211-217. [crossref]
- Villeponteau B, Cockrell R, Feng J (2000) Nutraceutical interventions may delay aging and the age-related diseases. Exp Gerontol 35: 1405-1417. [crossref]
- Socci DJ, Crandall BM, Arendash GW (1995) Chronic antioxidant treatment improves the cognitive performance of aged rats. Brain Res 693: 88-94. [crossref]
- Arzi A, Hemmati AA, Razian A (2004) Effect of vitamins C and E on cognitive function in mouse. Pharmacol Res 49: 249-252. [crossref]
- Linnoila M, Eckhardt M, Durcan M, Lister R, Martin P (1987) Interactions of serotonin with ethanol: Clinical and animal studies. Psychopharmacol Bull 452–457. [crossref]
- Dulawa SC, Holick KA, Gundersen B, Hen R (2004) Effects of chronic fluoxetine in animal models of anxiety and depression. Neuropsychopharmacology 29: 1321-1330. [crossref]
- Hascoët M, Bourin M, Colombel MC, Fiocco AJ, Baker GB (2000) Anxiolytic-Like Effects of Antidepressants after Acute Administration in a Four-Plate Test in Mice. Pharmacol Biochem Behav 65: 339-344. [crossref]
- Salchner P, Singewald N (2002) Neuroanatomical substrates involved in the anxiogenic-like effect of acute fluoxetine treatment. Neuropharmacol 43: 1238–1248. [crossref]
- Sinclair LI, Christmas DM, Hood SD, Potokar JP, Robertson A, et al. (2009) Antidepressant-induced jitteriness/anxiety syndrome: systematic review. Br J Psychiatry 194: 483-490. [crossref]
- Browning M, Reid C, Cowen PJ, Goodwin GM, Harmer CJ (2007) A single dose of citalopram increases fear recognition in healthy subjects. J Psychopharmacol 21: 684-690. [crossref]
- Wong WM, Hasemann S, Schwarz M, Zill P, Koller G, et al. (2008) Citalopram neuropharmacological challenge in alcohol-dependent patients and controls: pharmacogenetic, endocrine and psychobehavioral results. Pharmacopsychiatry 41: 72-78. [crossref]
- Serretti A, Calati R, Goracci A, Di Simplicio M, Castrogiovanni P, et al. (2010) Antidepressants in healthy subjects: what are the psychotropic/psychological effects? Eur Neuropsychopharmacol 20: 433-453. [crossref]
- Beneytez ME, LópezRodríguez ML, Rosado ML, Morcillo MJ, Orensanz L, et al. (1998) Preclinical pharmacology of B-20991, a 5-HT1A receptor agonist with anxiolytic activity. Eur J Pharmacol 344: 127-135. [crossref]
- Fletcher A, Forster EA, Bill DJ, Brown G, Cliffe IA, et al. (1996) Electrophysiological, biochemical, neurohormonal and behavioural studies with WAY-100635, a potent, selective and silent 5-HT1A receptor antagonist. Behav Brain Res 73: 337-353. [crossref]
- Bagdy G, Graf M, Anheuer ZE, Modos EA, Kantor S (2001) Anxiety-like effects induced by acute fluoxetine, sertraline or m-CPP treatment are reversed by pretreatment with the 5-HT2C receptor antagonist SB-242084 but not the 5-HT1A receptor antagonist WAY-100635. J Neuropsychopharmacol 4: 399–408. [crossref]
- Mefford IN, Oke AF, Adams RN (1981) Regional distribution of ascorbate in human brain. Brain Res 212: 223-226. [crossref]
- Harrison FE, May JM (2009) Vitamin C function in the brain: vital role of the ascorbate transporter SVCT2. Free Radic Biol Med 46: 719-730. [crossref]
- Glynn IM (1963) Transport adenosinetriphosphatase' in electric organ. The relation between ion transport and oxidative phosphorylation. J Physiol 169: 452-465. [crossref]
- Inagaki C (1970) Inhibition of Mg- and Mg, Na, K-activated adenosine tgriphosphatase activity by L-ascorbic acid and L-cysteine. Jpn J Pharmacol 20: 52-60. [crossref]
- Frey M, Pitts BJ, Askari A (1973) Vitamin C-effects on the Na,-K-adenosine triphosphate phosphohydrolase complexes of several tissues. Biochem. Pharmacol 22: 9–15. [crossref]
- Porsolt RD, Bertin A, Jalfre M (1977) Behavioral despair in mice: a primary screening test for antidepressants. Arch Int Pharmacodyn Ther 229: 327-336. [crossref]
- Borsini F, Meli A (1988) Is the forced swimming test a suitable model for revealing antidepressant activity? Psychopharmacol 94: 147–160. [crossref]
- Kelliher P, Kelly JP, Leonard BE, Sanchez C (2003) Effects of acute and chronic administration of selective monoamine re-uptake inhibitors in the rat forced swim test. Psychoneuroendocrinol 28: 332–347. [crossref]
- Griebel G, Moreau JL, Jenck F, Mutel V, Martin JR, et al. (1994) Evidence that tolerance to the anxiogenic-like effects of mCPP does not involve alteration in the function of 5-HT(2C) receptors in the rat choroid plexus. BehavPharmacol 5: 642-645. [crossref]
- Treit D, Fundytus M (1988) Thigmotaxis as a test for anxiolytic activity in rats. Pharmacol Biochem Behav 31: 959-962. [crossref]
- Gentsch C, Lichtsteiner M, Feer H (1987) Open field and elevated plus-maze: a behavioural comparison between spontaneously hypertensive (SHR) and Wistar-Kyoto (WKY) rats and the effects of chlordiazepoxide. Behav Brain Res 25: 101-107. [crossref]
- Schmitt U, Hiemke C (1998) Combination of open field and elevated plus-maze: a suitable test battery to assess strain as well as treatment differences in rat behavior. Prog Neuropsychopharmacol Biol Psychiatry 22: 1197-1215. [crossref]
- Choleris E, Thomas AW, Kavaliers M, Prato FS (2001) A detailed ethological analysis of the mouse open field test: effects of diazepam, chlordiazepoxide and an extremely low frequency pulsed magnetic field. Neurosci Biobehav Rev 25: 235–260. [crossref]
- Simon P, Dupuis R, Costentin J (1994) Thigmotaxis as an index of anxiety in mice. Influence of dopaminergic transmissions. Behav Brain Res 61: 59-64. [crossref]
- Van der Staay FJ, Van Nies J, Raaijmakers W (1990) The effects of aging in rats on working and reference memory performance in a spatial holeboard discrimination task. Behav Neural Biol 53: 356-370. [crossref]
- Bronikowski AM, Carter PA, Swallow JG, Girard IA, Rhodes JS, et al. (2001) Open-field behavior of house mice selectively bred for high voluntary wheel-running. Behav Genet 31: 309-316. [crossref]
- Johansson C, Ahlenius S (1989) Evidence for the involvement of 5-HT1A receptors in the mediation of exploratory locomotor activity in the rat. J Psychopharmacol 3: 32-35. [crossref]




