Purpose: This study examined patients who had a DDDR pacemaker and were exposed to different degrees of atrial and ventricular pacing. We aimed to determine the relationships between baseline features and percentages of atrial pacing (%AP), and ventricular pacing (%VP) in the first month post implant and subsequent onset of atrial fibrillation (AF) (primary outcomes). We also aimed to determine the relationships between time-dependent changes in %AP and %VP and AF recurrence (secondary outcomes).
Methods: Three hundred six patients were evaluated every 6 months up to 3 years of follow-up. The %AP and %VP were assessed at each follow-up visit.
Results: The mean time to the first episode of AF (n=227) was 15.3 ± 10.7 months. At the end of 3 years of follow-up, 74.2% patients developed AF events. At baseline, the mean %AP was significantly higher inpatients who subsequently developed AF than in those who did not (68.5% ± 19.7% versus33.3% ± 23.2%, P<0.0001). Similarly, mean %VP was significantly higher in patients who developed AF than in those without AF events (25.8% ± 14.5% versus 14.4% ± 11.9%, P<0.0001). Kaplan–Meier survival analysis demonstrated a significant increased risk of AF events in patients who subsequently developed AF compared with those who did not (log-rank test, P<0.0001).
Conclusions: Our study shows an association between %AP and %VP at baseline and time to appearance of the first AF episode after pacemaker implantation. During 3 years of follow-up, we also observed a significantly increased risk of AF events in the high %AP and high %VP groups at baseline.
sinus node disease, sick sinus syndrome, pacemaker, atrial fibrillation, pacing, left atrial diameter
AF: atrial fibrillation, AMS: auto mode switch, AP: atrial pacing, %AP: percentage of atrial pacing, AT: atrial tachycardia, AUC: area under the curve, AV: atrioventricular, LAD: left atrial diameter, LVEF: left ventricular ejection fraction, ROC: receiver operator characteristic, SND: sinus node disease, SSS: sick sinus syndrome, VP: ventricular pacing, %VP: percentage of ventricular pacing
Sinus node dysfunction is characterized by one or more of the following electrocardiographic manifestations: sinus bradycardia, sinus arrest, sino-atrial block, and supraventricular tachycardia alternating with sinus bradycardia and asystole (Brady-tachycardia syndrome) [1]. The presence of palpitations, dizziness, pre-syncope, or syncope associated with these electrocardiographic changes defines sinus node disease (SND). SND occurs more frequently in women than in men, resulting in a high morbidity between 60 and 69 years [2,3]. The most common form of SND has no clear etiology and is considered idiopathic or primary [4,5]. Paroxysmal atrial fibrillation (AF) is common in patients with sick sinus syndrome (SSS) and pacemakers, and leads to morbidity and an increased risk of stroke or death [6].
The recent DANPACE studyenrolled patients with SSS with no previous history of AF who received DDDR pacemakers [7,8], and aimed to determine the effect of atrial pacing on the occurrence of AF. This study did not find an association between increased atrial pacing and development of AF [7,8].
The present study examined patients who had a DDDR pacemaker and were exposed to different degrees of atrial pacing (AP) and ventricular pacing (VP). We aimed to determine the relationships between baseline features and percentages of atrial pacing (%AP) and ventricular pacing (%VP) in the first month postimplant and subsequent onset of AF (primary outcomes).Moreover, we aimed to determine the relationships between time-dependent changes in %AP and %VP and AF recurrence (secondary outcomes), thus allowing to treat the disease at earlier stages and change the natural course of this and its possible complications.
Study design
The present observational, prospective study was conducted at the Department of Cardiac Pacing and Cardiac Surgery of the Hospital e Clínica São Gonçalo. A cohort of patients received standard therapy for treatment of SND and DDDR pacemaker implantation. Follow-up was 3 years after the implantation procedure. The study inclusion criteria were as follows: (i) patients did not have electrocardiogram-documented AF or a previous history of paroxysmal AF; (ii) patients provided documentation of no cardiac ischemia before pacemaker implantation as proven by a myocardial scintigraphy at rest and during stress, a cardiac magnetic resonance imaging at rest and during stress, or pharmacological stress echocardiography; (iii) patients had a left ventricular ejection fraction (LVEF) ≥50% as measured by echocardiography; (iv) tests showing that the patients had SND (symptomatic bradycardia; documented sino-atrial block or sinus arrest with pauses >3 s or sinus bradycardia <40 bpm for >1 min while awake) before pacemaker implantation; and (v) tests showing that patients had no second- or third-degree atrioventricular (AV) block, and a PR interval <240 ms before pacemaker implantation.
Exclusion criteria were as follows: (i) ischemic heart disease; (ii) an LVEF <50%; (iii) heart valvar disease that may lead to AF; and (iv) symptoms suggestive of AF.
Enrolment of patients started in January 2009 and was terminated in September 2012. Patients were followed up until September 2015, and they were identified at our offices. The study was conducted in accordance with the Helsinki Declaration and was approved by the Ethics Committee of our hospital. All of the patients gave written informed consent before inclusion.
Implantation and programing of pacemakers
As a routine practice in our department, bipolar leads were implanted in the appendage of the right atrium and in the high septal region of the right ventricle. DDDR pacemakers from St. Jude Medical (St. Jude Medical, St. Paul, Minnesota, USA) and Medtronic (Medtronic, Palo Alto, CA, USA) were used. The rate adaptive function was activated in all of the pacemakers and programed with a lower rate of 60 bpm and an upper rate of 120 bpm In all of the pacemakers, we programed the paced atrioventricular interval to 140–220 ms and turned on the AV delay management algorithm that automatically searches for intrinsic conduction to prevent unnecessary right ventricular pacing. The maximum tracking rate was individualized and the auto mode switch (AMS) function was activated. AMS occurred when the atrial rate exceeded 170–180 bpm for a specific number of beats or period of time. The atrial tachycardia/atrial fibrillation (AT/AF) diagnostic suite provided detailed historical data, allowing us to identify and evaluate therapy for improved management of patients. Atrial sensitivity was programed to 0.5 mV.
Definition of atrial fibrillation
AF was defined as at least one episode of atrial irregular activity recorded by the atrial channel lasting ≥30 s.
Patients’ follow-up
Patients were evaluated 15 days after pacemaker implantation to assess the pocket, the site of the surgical incision, and to adjust the programing of the pacemaker. Fifteen days later, the patients returned for reassessment (1 month after pacemaker implantation). Data were obtained from the pacemaker at 1 month postimplant. Thereafter, patients were assessed every 6 months up to 3 years of follow-up. At each follow-up visit, we obtained a record (stored on a USB stick and then transferred to a computer) of the pacemaker memory data that had accumulated since the previous resetting of the memory. The %AP and %VP at each follow-up were calculated by the pacemaker using the number of paced and sensed beats. The occurrence and duration of AMS events were recorded. The onset of the first AF episode was also registered in each patient’s data record. Time to AF onset was defined as the number of days from baseline to the first recorded episode of AF lasting ≥30 s. Patients were censored due to death, loss to follow-up, or 3 years post-implant. According to our standard of care, all of the patients underwent echocardiography at baseline and annually after pacemaker implantation.
Statistical analysis
All enrolled patients were included in the analyses. Variables were compared between two groups using analysis of variance (ANOVA) for continuous measures and the χ2 test or Fisher’s exact test for categorical variables. The Kaplan–Meier method was used to describe the freedom from AF events over time, and differences between groups were assessed by the log-rank test. Baseline covariates and disease characteristics related to AF recurrence (P<0.1) were included in the Cox proportional hazards regression model, along with baseline %AP, %VP, and left atrial diameter (LAD). Forward selection was used to reduce the number of covariates. Receiver operator characteristic (ROC) curves were used to test the effectiveness of %AP and %VP at baseline in predicting AF events, and the area under the curve (AUC) was derived from the ROC curve. All statistical tests were two-tailed, and a P value of <0.05 was considered to indicate statistical significance. All statistical analyses were performed using SPSS v 18.0.
Patients
We screened 391 patients who received a DDD pacemaker to treat SND for preliminary inclusion (Figure 1). Among these, 85 patients were excluded because they fulfilled the following exclusion criteria: ischemic heart disease (n=21); an LVEF <50% (n=33); and symptoms suggestive of AF (n=31). The remaining 306 patients who adhered to the inclusion criteria were finally enrolled in the study (group 1: patients who did not develop AF, n=79; group 2: patients who developed AF, n=227). The baseline characteristics of patients in both groups are shown in Table 1.
Figure 1. Flow chart of the study. Enrolment, stratification, and follow-up of the patients are shown
Table 1. Baseline features of included patients
Variables |
Overall n=306 |
Group 1
n=79 |
Group 2 n=227 |
Pvalue |
Age (years) |
62.3 ± 13.2 |
61.8 ± 8.8 |
65.3 ± 12.4 |
0.0194 |
Sex, male, n(%) |
173 (56.5) |
42 (53.2) |
131 (57.7) |
0.5116 |
Hypertension, n (%) |
139 (45.4) |
16 (20.3) |
123 (54.2) |
<0.0001 |
CADa, n (%) |
31 (10.1) |
8 (10.1) |
23 (10.1) |
1.000 |
Diabetes mellitus, n (%) |
132 (43.1) |
22 (27.8) |
110 (48.5) |
0.0038 |
ACEI or ARB, n (%) |
125 (40.8) |
14 (17.7) |
111 (48.9) |
<0.0001 |
Diuretics, n (%) |
45 (14.7) |
3 (3.8) |
42 (18.5) |
0.0008 |
Ca++ channel blockers, n (%) |
46 (15.0) |
2 (2.5) |
44 (19.4) |
<0.0001 |
Beta-blockers, n (%) |
29 (9.5) |
7 (8.9) |
22 (9.7) |
1.0000 |
AP (%) |
59.4 ± 25.7 |
33.3 ± 23.3 |
68.5 ± 19.7 |
<0.0001 |
VP (%) |
23.4 ± 17.9 |
14.4 ± 11.9 |
26.6 ± 18.6 |
<0.0001 |
LAD (cm) |
3.9 ± 0.4 |
3.6 ± 0.2 |
4.0 ± 0.4 |
0.6161 |
LVEF (%) |
64.2 ± 7.3 |
63.8 ± 7.4 |
64.4 ± 7.3 |
0.9996 |
ACEI: angiotensin-converting enzyme inhibitor; AP: atrial pacing; ARB: angiotensin-receptor blocker; CAD: coronary artery disease; LAD: left atrial diameter; LVEF: left ventricular ejection fraction; VP: ventricular pacing.
aPatients with coronary artery disease had undergone coronary angiography or computed tomography angiography of coronary arteries at some time in their life. This indicated that they had some degree of obstruction in the coronary arteries, but this did not cause ischemia.
The %AP and %VP at baseline compared with the probability of AF
At baseline, mean %AP was significantly higher in patients who subsequently developed AF compared with those who did not (33.3% ± 23.2% versus 68.5% ± 19.7%, P<0.0001). Similarly, mean %VP was significantly higher in patients who developed AF compared with those who did not (25.8% ± 14.5% versus 14.4% ± 11.9%, P<0.0001).
AF events
The mean time to the first episode of AF (n=227) was 15.3 ± 10.7 months. At the end of 3 years of follow-up, 25.8% of patients did not have AF episodes, while 74.2% of patients developed AF events.
ROC analysis showed that the %AP and %VP at baseline were feasible and effective predictors for the long-term risk of AF episodes (AUC: 0.8828, 95% confidence interval: 0.8267–0.9390, P<0.0001; AUC: 0.7484, 95% confidence interval: 0.6836-0.8131, P<0.0001, respectively). The potential cut-off points of %AP and %VP derived from ROC analysis were 45.5% (sensitivity: 91.63%; specificity: 81.01%) and 18.5% (sensitivity: 68.72%; specificity: 72.15%), respectively (Figure 2A and B). Using these thresholds, Kaplan–Meier survival analysis demonstrated a significant increased risk of AF events in the high %AP group (>45.5%) and in the high %VP group (>18.5%) (both P<0.0001, log-rank test, Figure 3A and B, respectively).
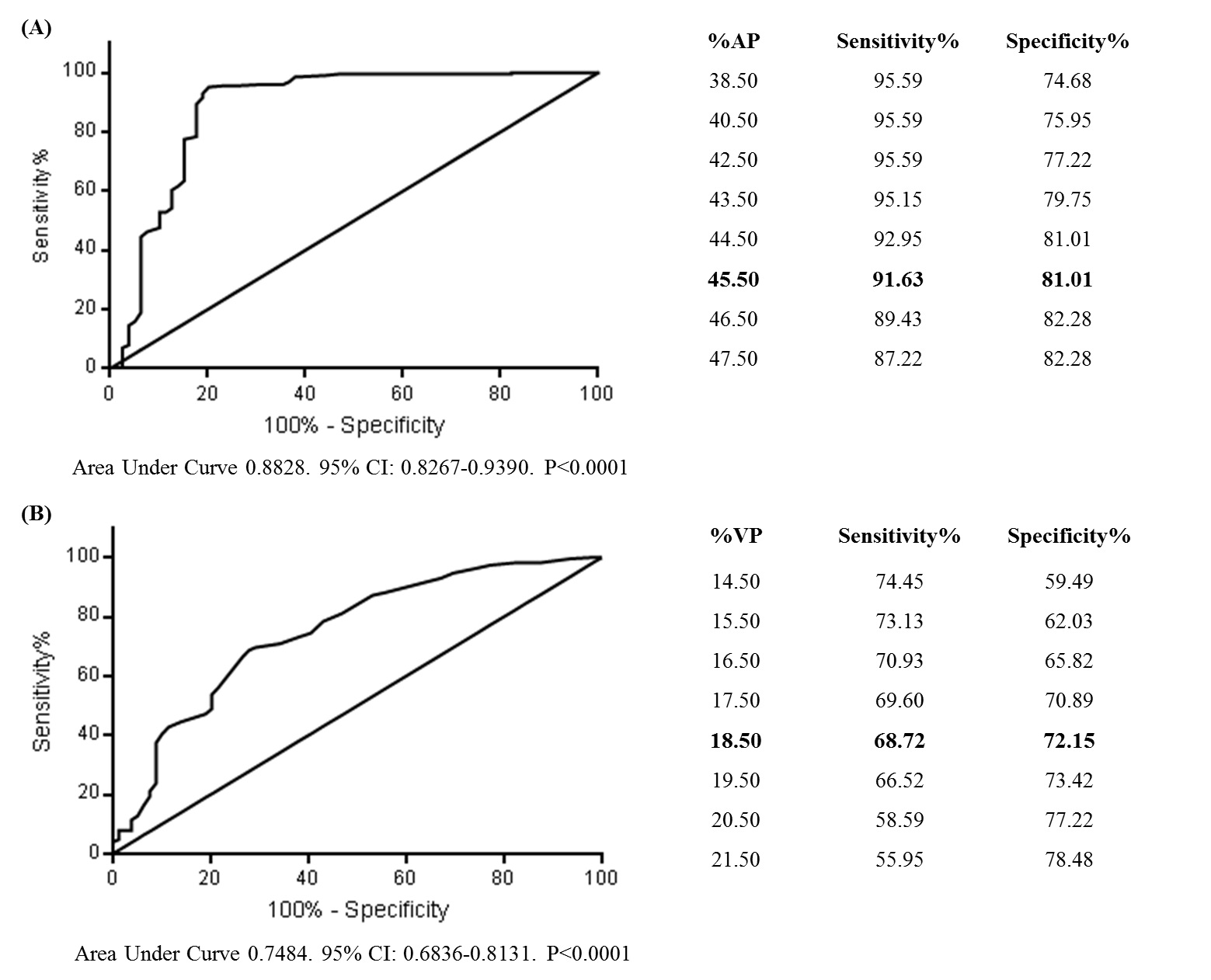
Figure 2. Receiver operating characteristic analysis testing the predictive ability of (A) %AP and (B) %VP at baseline for atrial fibrillation episodes. CI: confidence interval
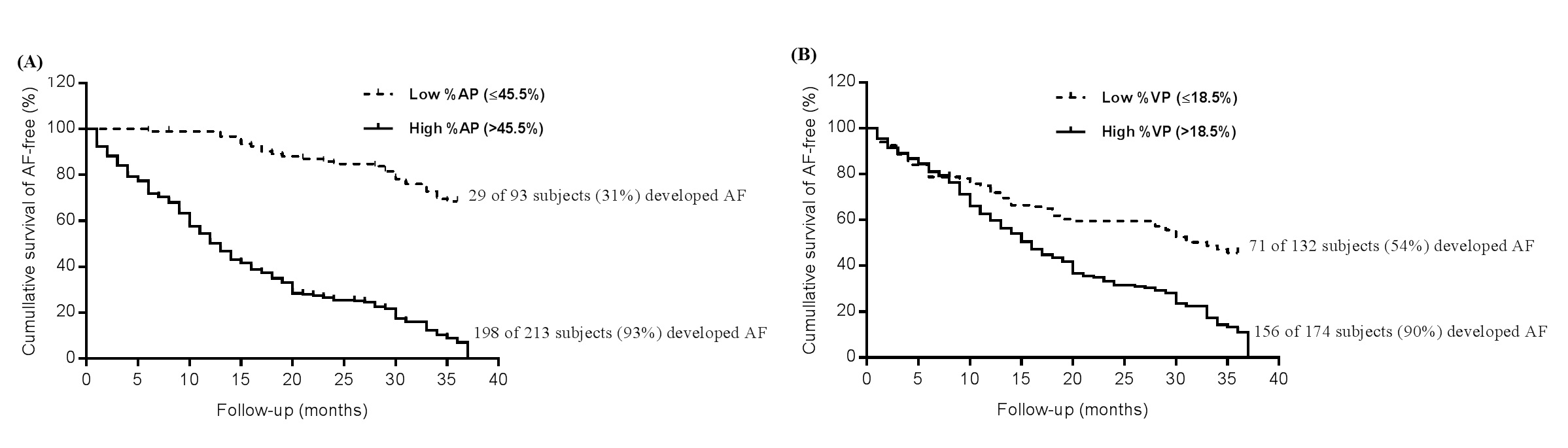
Figure 3. Kaplan–Meier analysis for AF events in (A) low and high %AP, and (B) in low and high %VP at baseline (both P<0.0001). AF: atrial fibrillation; AP: atrial pacing; VP: ventricular pacing
The %AP and %VP, left atrial diameter, and LVEF during follow-up
Throughout follow-up, we did not observe changes in %AP %VP, and LVEF in both groups. However, LAD showed reduction in group 1 in comparison to group 2 at specific time points (Table 2).
Table 2. %AP and %VP during 3 years of follow up.
%AP |
Baseline |
6 months |
12 months |
18 months |
24 months |
30 months |
36 months |
All P-value |
Overall |
59.42 ± 25.72 |
60.38 ± 25.73 |
60.84 ± 25.80 |
61.21 ± 26.06 |
61.74 ± 26.13 |
62.04 ± 26.31 |
62.53 ± 26.31 |
0.8041 |
Group 1 |
33.33 ± 23.15 |
33.86 ± 23.13 |
33.87 ± 22.94 |
34.01 ± 23.18 |
34.51 ± 23.5 |
34.14 ± 23.62 |
34.34 ± 23.60 |
˃0.9999 |
Group 2 |
68.51 ± 19.67 |
69.61 ± 19.43 |
70.23 ± 19.33 |
70.68 ± 19.56 |
71.22 ± 19.54 |
71.74 ± 19.36 |
72.34 ± 19.16 |
0.4066 |
Δ group vs. group 2 |
35.18 |
35.75 |
36.36 |
36.67 |
36.71 |
37.61 |
38.00 |
˂0.0001 |
%VP |
Baseline |
6 months |
12 months |
18 months |
24 months |
30 months |
36 months |
All P-value |
Overall |
23.43 ± 17.92 |
23.04 ± 14.84 |
23.07 ± 14.65 |
22.87 ± 14.36 |
22.69 ± 14.08 |
22.30 ± 14.07 |
22.41± 14.03 |
0.9719 |
Group 1 |
14.41 ± 11.87 |
14.30 ± 11.64 |
14.30 ± 11.33 |
14.11 ± 10.86 |
14.16 ± 10.56 |
14.10 ± 10.46 |
14.51 ± 10.70 |
˃0.9999 |
Group 2 |
26.57 ± 18.61 |
26.08 ± 14.65 |
26.11 ± 14.47 |
25.92 ± 14.19 |
25.65 ± 13.96 |
25.15 ± 14.06 |
25.16 ± 14.03 |
0.9479 |
Δ group vs. group 2 |
12.16 |
11.78 |
11.81 |
11.80 |
11.49 |
11.05 |
10.66 |
˂0.0001 |
LAD(cm) |
Baseline |
12 months |
24 months |
36 months |
All P- value |
Overall |
3.86±0.42 |
4.05±2.51 |
3.95±0.45 |
4.08 ± 1.83 |
0.3023 |
Group 1 |
3.59±0.22 |
3.59±0.22 |
3.60±0.22 |
3.61 ± 0.22 |
0.8993 |
Group 2 |
3.96±0.43 |
4.22±2.90 |
4.07±0.45 |
4.24 ± 2.10 |
0.3068 |
group 1 vs. group 2 |
0.3699 |
0.6243* |
0.46910 |
0.6256* |
*<0.05 |
LEVF(%) |
Baseline |
12 months |
24 months |
36 months |
All P- value |
Overall |
64.22 ± 7.34 |
64.19 ± 7.33 |
64.93 ± 13.95 |
64.28 ± 7.35 |
0.7293 |
Group 1 |
63.75 ± 7.41 |
63.77 ± 7.41 |
66.47 ± 24.50 |
64.04 ± 7.25 |
0.5430 |
Group 2 |
64.38 ± 7.32 |
64.33 ± 7.31 |
64.4 ± 7.32 |
64.36 ± 7.39 |
0.9997 |
group 1 vs. group 2 |
0.6364 |
0.5582 |
2.070 |
0.3249 |
0.7014 |
AP: atrial pacing, LAD: left atrial diameter, LVEP: left ventricular ejection frction, VP: ventricular pacing
Adjustment for several possible confounders known to be associated with AF
As shown in Table 3, age, hypertension, diabetes, use of angiotensin-converting enzyme inhibitor/angiotensin-receptor blocker (ACEI/ARB), diuretics, calcium channel blockers (CCB), %AP, and %VP were significantly different (defined as P<0.1) between the groups. We included these variables plus LAD and LVEF (they are both outcome-relevant measurements) in the Cox multivariate logistic regression model by the forward stepwise (Wald) method. Male sex, hypertension, %AP, %VP, and LAD were risk factors of development of AF (Table 4).
Table 3. Cox proportional hazards model to examine the associations between variables and AF events.
Variables |
OR |
95%CI |
Pvalue |
Age |
0.995 |
0.981–1.009 |
0.469 |
Male sex |
1.363 |
1.019–1.822 |
0.037 |
Hypertension |
2.062 |
1.018–4.178 |
0.044 |
Diabetes mellitus |
0.949 |
0.705–1.277 |
0.729 |
Coronary artery disease |
0.782 |
0.320–1.910 |
0.590 |
%Atrial pacing |
1.030 |
1.022–1.038 |
<0.001 |
%Ventricular pacing |
1.021 |
1.012–1.029 |
<0.001 |
Left ventricular ejection fraction |
0.993 |
0.974–1.013 |
0.501 |
Left atrial diameter |
11.110 |
6.209–19.879 |
<0.001 |
ACEI/ARB |
0.911 |
0.477–1.738 |
0.776 |
Diuretic |
0.954 |
0.646–1.411 |
0.815 |
Calcium channel blocker |
1.093 |
0.741–1.613 |
0.654 |
Β-blocker |
0.994 |
0.393–2.517 |
0.991 |
ACEI: angiotensin-converting enzyme inhibitor; AF: atrial fibrillation; ARB: angiotensin-receptor blocker; OR: odds ratio; 95%CI: 95% confidence interval.
Table 4. Cox proportional hazards model by the forward stepwise (Wald) method to examine the associations between variables and AF events.
Variables |
OR |
95%CI |
Pvalue |
Male sex |
1.341 |
1.016–1.770 |
0.038 |
Hypertension |
1.941 |
1.458–2.584 |
<0.0001 |
%Atrial pacing |
1.028 |
1.021–1.035 |
<0.001 |
%Ventricular pacing |
1.021 |
1.013–1.029 |
<0.001 |
Left atrial diameter |
11.059 |
6.550–18.673 |
<0.001 |
AF: atrial fibrillation; OR: odds ratio; 95%CI: 95% confidence interval.
In the present study, we found associations of %AP and %VP at baseline, as well as male sex, hypertension, and higher left atrial diameter with the time to appearance of the first AF episode after pacemaker implantation. We also found that by using the thresholds from the ROC curve, Kaplan–Meier survival analysis showed a significant increased risk of AF events in the high %AP and high %VP groups.
Elkayam et al. [9] reported that AP and VP in patients with SND were predisposing factors for development of AF. However, the DANPACE trial showed that in 396 patients who received DDDR pacemakers because of SSS, 72% developed AF as indicated by AMS episodes at some point during follow-up [8]. This trial also showed no association between %AP and development of AF [8].
We found that the mean time for the first AF episode after pacemaker implantation was 15.3 ± 10.7 months, and among 306 patients, 74% developed AF. The risk of stroke in patients with AF can be evaluated by using the CHADS2 and the CHA2DS2-VASc score system. Svendsen et al. hypothesized that these risk scores and their individual components can also be applied to patients paced for SSS to evaluate the risk of stroke and death [10]. They showed that CHADS2 and CHA2DS2-VASc scores were associated with an increased risk of stroke and death in patients paced for SSS, irrespective of the presence of AF. In our study, the mean %AP and %VP in patients who did not develop AF were lower than those in patients who showed AF events during the follow-up period. In patients without AF, the %AP was 34% and the %VP was 14% at the 36th month of follow-up. In patients that developed AF, the %AP was 72% and the %VP was 25% at the 36th month of follow-up. The variation (∆) between these two groups was 38% for %AP (72%−34%) and 9% for %VP (25%−14%) at the 36th month of follow-up. Furthermore, ROC analysis showed that %AP and %VP were feasible and effective predictors for long-term risk of AF episodes. The potential cut-off points of %AP and %VP derived from ROC analysis were 45.5% and 18.5%, respectively. These thresholds can be important because they may allow identification of potential individuals who will develop AF. This enables patients to be more closely and routinely followed up. AP is considered potentially pro-arrhythmogenic by some scientists, while others have focused on atrial overdrive pacing strategies for preventing AF. Results from studies on overdrive pacing have not shown a convincing benefit in prevention of AF, and these trials only had modest sample sizes [11,12]. The ASSERT trial with 2580 patients did not demonstrate any benefit of continuous atrial overdrive pacing [13].
Pacing in the right atrium may induce atrial dyssynchrony and subsequent AF [14]. The percentage of atrial pacing can lead to hemodynamic alterations, and changes in the shape and velocity of intra-atrial, interatrial, and atrioventricular conduction, as well as alter cellular properties. An association between %AP and AF may be possible because of differences among baseline variables. Theoretically, a lower intrinsic heart rate may indicate a greater severity of SSS and thus be associated with a higher %AP and a risk of developing AF, without implications of a causal connection. The rate response to be associated with a higher degree of AP, it also theoretically possible, that this feature is another contributing factor. Because all of the patients in our study had this feature turned on, we are not able to confirm this possibility. Pacing strategies targeting the intra- and interatrial conduction tracts [15,16] and algorithms aimed at minimizing atrial pacing should be further examined.
The mechanism for the increased incidence of AF with VP remains unclear, but hemodynamics are thought to be involved. Right VP leads to left ventricular remodeling, increases mitral regurgitation, and modestly reduces ejection fraction [17]. Furthermore, changing the relationship between atrial and ventricular timing, as can occur with VP, increases atrial pressure and causes stretch-related changes. This may increase the incidence of AF [18].
2021 Copyright OAT. All rights reserv
In our study, male sex, hypertension, and a higher LAD appeared to be associated with onset of AF, despite using antihypertensive drugs, including ACEI/ARB. A higher LAD might be due to increased intracavitary pressure and possible scar tissue areas on the left atrium. Furthermore, the state of sympathetic overactivity present in hypertension may contribute to triggering factors involved in AF.
A limitation of this study is that patients who were included in the study could have presented with an episode of asymptomatic AF. However, this was unable to be determined because they did not have an implantable looper.
The occurrence of AF may sometimes lead to under-sensing in the atrium and thus inappropriate AP. The use of beta-blockers and the onset of antiarrhythmic drugs after the first AF event may inhibit the sinus node and thus cause an increase in %AP. However, we were unable to satisfactorily adjust for beta-blocker or antiarrhythmic drug use because of the heterogeneity of drugs and doses used, and their wide variation of effects in patients. Some comorbid conditions, such as hypertension, diabetes, and a high degree of sinus dysfunction, more common in one of the groups, led to a higher percentage of atrial pacing and probably to onset of AF. Furthermore, the causality of atrial pacing and development of AF was not able to be determined in this study.
The present study shows associations of %AP and %VP at baseline, male sex, hypertension, and higher left atrial diameter with the time to appearance of the first AF episode after pacemaker implantation. We also observed a significant increased risk of AF events in the high %AP and high %VP groups during 3 years of follow-up.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study
The study was sponsored by health insurance plans of the state of Rio de Janeiro and Pacemed.
The authors declare that they have no conflict of interest.
The authors thank all the participants in this study and Pacemed for the technical support. The study was sponsored by health insurance plans of the state of Rio de Janeiro and Pacemed.
- Ferrer MI (1968) The sick sinus syndrome in atrial disease. JAMA 206: 645-646. [Crossref]
- Shaw DB, Holman RR, Gowers JI (1980) Survival in sinoatrial disorder (sick-sinus syndrome). Br Med J 280: 139-141. [Crossref]
- de Marneffe M, Gregoire JM, Waterschoot P, Kestemont MP (1993) The sinus node function: normal and pathological. Eur Heart J 14: 649-654. [Crossref]
- Shaw DB (1976) The etiology of sino-atrial disorder (Sick Sinus Syndrome). Am Heart J 92: 539-540. [Crossref]
- Nieminski KE, Kay RH, Rubin DA (1984) Current concepts and management of the sick sinus syndrome. Heart Lung 13: 675-681. [Crossref]
- Glotzer TV, Hellkamp AS, Zimmerman J, Sweeney MO, Yee R, et al. (2003) MOST Investigators (2003). Atrial high rate episodes detected by pacemaker diagnostics predict death and stroke: report of the Atrial Diagnostics Ancillary Study of the MOde Selection Trial (MOST). Circulation 107: 1614–1619.
- Nielsen JC, Thomsen PE, Højberg S, Møller M, Vesterlund T, et al. (2011) A comparison of single-lead atrial pacing with dual-chamber pacing in sick sinus syndrome. Eur Heart J 32: 686-696. [Crossref]
- Hjortshøj S, Riahi S, Nielsen JC, Skjøth F, Lundbye-Christensen S, et al. (2014) DANPACE Investigators. Does atrial pacing lead to atrial fibrillation in patients with sick sinus syndrome? Insights from the DANPACE trial. Europace 16: 241–245.
- Elkayam LU, Koehler JL, Sheldon TJ, Glotzer TV, Rosenthal LS, et al. (2011) The influence of atrial and ventricular pacing on the incidence of atrial fibrillation: a meta-analysis. Pacing Clin Electrophysiol 34: 1593-1599. [Crossref]
- Svendsen JH, Nielsen JC, Darkner S, Jensen GV, Mortensen LS, et al. (2013) CHADS2 and CHA2DS2-VASc score to assess risk of stroke and death in patients paced for sick sinus syndrome. Heart 99: 843-848. [Crossref]
- Carlson MD, Ip J, Messenger J, Beau S, Kalbfleisch S, Gervais P, et al. (2003) Atrial Dynamic Overdrive Pacing Trial (ADOPT) Investigators (2003) A new pacemaker algorithm for the treatment of atrial fibrillation: results of the Atrial Dynamic Overdrive Pacing Trial (ADOPT). J Am Coll Cardiol 42: 627–633.
- Nielsen JC, Kristensen L, Andersen HR, Mortensen PT, Pedersen OL, et al. (2003) A randomized comparison of atrial and dual-chamber pacing in 177 consecutive patients with sick sinus syndrome: echocardiographic and clinical outcome. J Am Coll Cardiol 42: 614–623.
- Xie JM, Fang F, Zhang Q, Chan JY, Yip GW, et al. (2012) Atrial dysfunction and interatrial dyssynchrony predict atrial high rate episodes: insight into the distinct effects of right atrial appendage pacing. J Cardiovasc Electrophysiol 23: 384–390.
- Andersen HR, Nielsen JC, Thomsen PE, Thuesen L, Mortensen PT, et al. (1997) Long-term follow-up of patients from a randomised trial of atrial versus ventricular pacing for sick-sinus syndrome. Lancet 350: 1210-1216. [Crossref]
- Dabrowska-Kugacka A, Lewicka-Nowak E, Rucinski P, Zagozdzon P, Raczak G, et al. (2009) Atrial electromechanical sequence and contraction synchrony during single- and multisite atrial pacing in patients with brady-tachycardia syndrome. Pacing ClinElectrophysiol 32: 591–603.
- Dabrowska-Kugacka A, Lewicka-Nowak E, Rucinski P, Kozlowski D, Raczak, G, et al. (2010) Single-site Bachmann’s bundle pacing is beneficial while coronary sinus pacing results in echocardiographic right heart pacemaker syndrome in brady-tachycardia patients. Circ J 74: 1308–1315.
- Yu CM, Chan JY, Zhang Q, Omar R, Yip GW, et al. (2009) Biventricular pacing in patients with bradycardia and normal ejection fraction. N Engl J Med 361: 2123-2134. [Crossref]
- Klein LS, Miles WM, Zipes DP (1990) Effect of atrioventricular interval during pacing or reciprocating tachycardia on atrial size, pressure, and refractory period. Contraction-excitation feedback in human atrium. Circulation 82: 60-68. [Crossref]



