Abstract
Background: The symptoms of upper gastrointestinal(GI) tract are often the subject of clinical consultation. Histopathology along with endoscopy remains the definitive method of diagnosing upper GI lesions. However there is a dearth of region specific data from Kerala.
Aim: To describe the histopathology of upper GI biopsies of a tertiary care hospital in Kerala and to correlate them with their endoscopic and clinical presentations.
Methods: This was a descriptive cross-sectional study in which 200 successive upper GI endoscopic biopsies were examined. The samples of tissues were treated and classified based on WHO guidelines. Other parameters observed were the H. pylori infection, intestinal metaplasia, atrophic changes, neutrophilic activity, parietal cell hyperplasia, and dysplastic changes. Data analysis was done using descriptive statistics and correlated with their patient records.
Results: The sample mainly included patients over 40 (80.5%), with a minor preponderance of males (1.2:1). The most common complaint was abdominal pain(48.5%), in those with antral gastritis (48.5%), and in the pyloric region (70.5%). About 70% of inflammatory lesions were attributable to chronic gastritis. Malignanneoplasms comprised 24%, which was most frequent. About 4.5% of samples had benign polyps, and 1.5% pre-malignant lesions. Helicobacter pylori was found in 30%cases of chronic gastritis, intestinal metaplasia was found in 7%, and dysplasia in 1.5%.
Conclusion: There was a higher incidence of inflammatory pathology, but malignancies represented a large share of cases in persons over 40 years. Early identification and management of pre-malignant changes and H. pylori infection highlight the importance of biopsy, emphasizing its urgency and regular follow up
Keywords
upper gastrointestinal tract, endoscopic biopsy, histopathology, chronic gastritis, helicobacter pylori, gastric adenocarcinoma
Abbreviations
EGD: Esophagogastroduodenoscopy; GERD: Gastroesophageal reflux disease; GI: Gastrointestinal; H&E: Hematoxylin & eosin; PAS: Periodic acid schiff; SCC: Squamous cell carcinoma; UGI: Upper gastrointestinal; WHO: World Health Orgnization
Introduction
One of the most common clinical manifestations that forces patients globally to visit healthcare facilities is upper gastrointestinal (UGI) symptoms, with the prevalence being estimated to be around 25 percent of patients who have to consult the medical institution each year [1]. Dyspepsia-related conditions, such as peptic ulcer disease, gastroesophageal reflux disease (GERD), and upper gastrointestinal neoplasms, continue to be a significant cause of gastrointestinal morbidity and mortality [2]. These manifestations are mainly common, with upper abdominal pain or discomfort being experienced by 8%–54% of groups, heartburn and regurgitation experienced by 10%–48% and 9%–45% of individuals respectively [3,4]. However, these clinical presentations are usually non-specific and overlap in a range of benign, premalignant and malignant entities. The correct interpretation of them requires the access to the environmental exposures, dietary pattern, and host factors, hence the need to have dependable diagnostic modalities.
One of the most important developments in the field of gastroenterology occurred in 1968 with the introduction of flexible fibre-optic endoscopy, which was the revolutionary form of examination in the case of oesophageal, gastric and duodenal disease. [5]. The workhorse when it comes to the investigation of persistence or alarm-related UGI symptoms is that of esophagogastroduodenoscopy (EGD). This is a minimally invasive surgery that allows a direct visualisation of the oesophagus, stomach and proximal duodenum, where indications of the procedure are discussed as the evaluation of dysphagia, gastrointestinal bleeding, refractory GERD, esophageal strictures, coeliac disease as well as unexplained diarrhea [6]. In addition to its diagnostic value, EGD allows direct visualisation allows the use of histopathological evaluation which is more sensitive to identifying mucosal pathology compared to imaging modalities [7,8]. It is particularly helpful in the detection of inflammatory diseases like oesophagitis, gastritis, and duodenitis and vascular malformations, Mallory-Weiss tears, and early neoplasia.
Although EGD would provide correct visualisation of the mucosal abnormalities, endoscopic impressions on their own might not be reliable in definitive diagnosis. Introduction of nonneoplastic or premalignant alterations may resemble malignancies and minor neoplastic lesions may become benign. Therefore, histopathology of biopsy samples is essential in differentiating inflammatory, benign, premalignant, and malignant ones [9]. Combined use of endoscopic and histopathological data is useful in the early detection, therapeutic decision making, and prognostic evaluation. It has been noted in numerous studies that there is a need to correlate endoscopic observations with histopathological verification in order to obtain diagnostic accuracy and the best management of patients [10–15].
Jonnalagadda, et al. [16] report that endoscopic biopsy and histopathology is one of the most effective modalities that can be used to diagnose neoplastic and non-neoplastic lesions, especially malignancies, which enhances clinical outcomes. Rauta and associates realised that non-neoplastic lesions are commonly diagnosed by increased use of histopathological diagnosis, despite the fact that malignant growths can be easily identified on endoscopy, which supports the diagnostic usefulness of endoscopy [17]. On the same note, Gunay and coauthors investigated the agreement between clinical and histopathological diagnosis and found that upper GI endoscopy is justified with at least one of the red flag symptoms, but empirical treatment could be used in younger patients who do not have the red flag symptoms [18]. These among other similar studies emphasise the significance of combining endoscopy and histopathological evaluation, particularly with the early detection of neoplastic change [19–21].
Though this is the case, there are still gaps that exist. A good part of what has been published in India and other neighbouring countries focuses on clinical or endoscopic appearances without correlating with histopathological diagnoses. Specifically, there are limited data on Kerala, despite the fact that the area has unique demographic and dietary patterns that can shape the manifestation of the disease. In addition, whereas the malignant lesions have been widely studied, there are less reports that describe the entire range of non-neoplastic and premalignant lesions that are essential in cancer prevention. Past research has also had limitations of small sample sizes or non-standardised histopathological parameters thus reducing comparability. By demonstrating such gaps, there is a need to incorporate region specific data that combine clinical presentation, endoscopic appearance, and histopathological observations to enhance the characterisation of upper GI pathology in the usual practice.
Considering these gaps, the current research was conducted to describe the histopathological range of upper GI biopsies in a tertiary care hospital in Kerala and to compare the results with the clinical presentation and endoscopic images. The study will produce region-specific data to improve the diagnosis of diseases earlier, make clinical decisions, and guide surveillance measures of non-neoplastic and neoplastic diseases in the upper GI by integrating demographic, clinical, endoscopic, and histopathological data.
Materials and methods
Study design
The research is a descriptive cross-sectional study that is conducted following the STROBE reporting guidelines [22]. The main purpose was to define the range of histopathological lesions, which were detected in upper gastrointestinal (GI) biopsies and to determine their relations with clinical symptoms and endoscopic changes.
Study setting and duration
It was conducted in the Department of Pathology in a tertiary referral centre in Kerala over a period of 1 year from November 2022 to November 2023. The endoscopy unit of the hospital serves a heterogeneous group of patients with patients across the central zone of Kerala, hence covering a wide range of upper GI disorders.
Ethical considerations
The study was commenced after getting approved by the Institutional Ethics Committee. The additional tissue section examination did not charge any financial strain on the patients. The entire process was carried out in line with the institutional and national ethical guidelines of biomedical research. Anonymisation of patient information was done and data was only used in an academic and clinical research. Informed consent was also taken according to institutional protocol during the time of biopsy.
Study population
The target population of the study consisted of all-aged and gender patients with upper GI symptoms and endoscopic biopsy during the study period. All the specimens obtained were of the esophagus, esophagogastric junction, stomach and proximal duodenum. The cases were not to be included in case tissues were not considered sufficient to carry out a histopathological test.
Sample size and sampling
The sample size was calculated using the formula n=4pq/d2. Drawing on the prevalence of malignant lesions reported by Veerendrasagar, et al. [23] a prevalence (p) of 0.68 was adopted, with q = 1 − p = 0.32 and a margin of error (d) of 10 % (p = 0.068). Replacement of these values produced n = 4* 0.68* 0.32/ 0.068 2 = 188; 200 was therefore selected as a sample size. Two hundred upper GI biopsy samples in a sequence that met the inclusion criteria were analysed and there was use of consecutive sampling to reduce selection biasness.
Data collection
Patient records were used to extract clinical and demographic data, such as age, sex and presenting symptoms. Contemporary findings of endoscopy including biopsy site and appearance of the macroscopic lesions were recorded. All data were keyed in a tabulated proforma designed especially to conduct this research.
Laboratory methods
Biopsy specimens were fixed in 10 % buffered formalin, underwent routine processing, and embedded in paraffin wax. Sections were cut at 4-5 micrometer thickness, following which staining with haematoxylin and eosin (H&E) was done in accordance with the conventional histopathological procedures [24]. Appropriate special stains were used: Helicobacter pylori Giemsa or Warthin-Starry and intestinal metaplasia periodic acid-schiff (PAS) or alcian blue [25-28]. Quality control was done using departmental policies such as verifying duplicate slides in instances of diagnostic doubt.
Variables and measures
The main outcome variable was the histopathological diagnosis of biopsy specimens and the explanatory variables were the patient demographics, presenting symptoms, endoscopic findings and the site of biopsy. The classification of lesions according to the world health organisation of tumours of the digestive system was inflammatory, benign neoplastic, premalignant, and malignant [29]. Inflammatory lesions were classified further as acute or chronic, or chronic active gastritis, intestinal metaplasia or dysplasia were defined as premalignant lesions, whereas malignant lesions were categorised using established histological criteria of upper GI tumours. Others that were noted were Helicobacter pylori infection, neutrophilic, atrophy, and parietal cell hyperplasia.
Statistical analysis
The information was recorded and stored in Microsoft Excel sheets. The descriptive statistics were used, and the categorical variables, including demographic characteristics, clinical symptoms, endoscopic results, and histopathological diagnoses were summarised with the help of frequencies and percentages. The continuous variables such as the age were represented in terms of means with standard deviations. Such variables as age group, sex, presenting symptoms (dysphagia, reflux, dyspepsia, abdominal pain, nausea/vomiting, anorexia/weight loss, haematemesis/melena), biopsy site, type of lesion, presence of Helicobacter pylori, and histological variables (intestinal metaplasia, neutrophilic activity, atrophy, parietal cell hyperplasia, dysplasia) were analysed. There were also categorisation and quantification of endoscopy findings. Findings were given in tables and graphical form which shows the distribution of these variables. Statistical tests were carried out in JAMOVI 2.5.3 at p 0.05.
Results
The study cohort comprised of 200 patients. Majority (80.5%) were aged above 40 years, with a mean age of 54.1 ± 12.2 years. The cohort was slightly male dominant with 111 males (55.5%) and 89 females (44.5%) producing a male to female ratio of 1.2:1.
The most frequent presenting symptom, reported by 97 patients (48.5%), was abdominal pain. This was followed by dyspepsia observed in 51 patients (25.5%), and dysphagia in 29 patients (14.5%). In 22 patients (11%) reflux was reported, in 34 patients (17%) nausea or vomiting, in 20 patients (10%) anorexia or weight loss, and in 18 patients (9%) haematemesis or melena. A hundred and thirty-five patients (67.5%), reported a single symptom, and 65 (32.5%), experienced the symptoms together.
The most common endoscopic finding was antral/perinatal gastritis, followed by ulceroproliferative growths. Only a few patients showed duodenitis or gastric outlet obstruction and a minority of patients had normal studies. Pylorus was the major location of biopsy, and oesophageal and duodenal samples were underrepresented (see Table 1).
Endoscopic finding |
n (%) |
Site of biopsy |
n (%) |
Category |
Diagnosis |
n (%) |
Antral/ periantral gastritis |
97 (48.5) |
Pylorus (including antrum/canal) |
141 (70.5) |
Inflammatory (70.0) |
Chronic gastritis |
139 (69.5) |
Ulceroproliferative growth |
44 (22.0) |
Body of stomach |
19 (9.5) |
|
Chronic inflammation |
3 (1.5) |
Normal study |
18 (9.0) |
Oesophagus |
18 (9.0) |
Benign neoplasms (4.5) |
Hyperplastic polyp |
6 (3.0) |
Polyp/nodule |
9 (4.5) |
Gastroesophageal junction |
11 (5.5) |
|
Fundic gland polyp |
2 (1.0) |
Submucosal swelling |
7 (3.5) |
Cardia |
1 (0.5) |
Tubular adenoma |
1 (0.5) |
Pangastritis |
6 (3.0) |
Fundus |
7 (3.5) |
Premalignant (1.5) |
Intestinal metaplasia/dysplasia |
2 (1.0) |
Atrophic gastritis |
3 (1.5) |
Duodenum (D1) |
3 (1.5) |
|
Barrett’s oesophagus with dysplasia |
1 (0.5) |
Biliary gastritis |
3 (1.5) |
|
|
Malignant (24.0) |
Gastric adenocarcinoma |
31 (15.5) |
Gastric outlet obstruction |
2 (1.0) |
|
|
Oesophageal squamous cell carcinoma (SCC) |
15 (7.5) |
Duodenitis |
1 (0.5) |
Carcinoma with neuroendocrine differentiation |
1 (0.5) |
|
Gastrointestinal stromal tumour (GIST) |
1 (0.5) |
Table 1. Additional histological features and clinico–pathological correlation (n = 200)
The histological findings were found to be mainly due to inflammatory lesions, which comprised 70 percent of all the biopsies, mostly representing chronic gastritis. Malignant lesions constituted 24% of cases, the most common type of which was gastric adenocarcinoma, and then the squamous cell carcinoma of the oesophagus. Benign neoplasms were isolated to gastric polyps (4.5%), and the premalignant changes were rare (1.5%), including intestinal metaplasia, dysplasia, and Barrett's oesophagus with dysplasia (see Table 2, Figures 1 and 2).
Category |
Diagnosis |
n (%) |
Inflammatory (70.0) |
Chronic gastritis |
139 (69.5) |
Chronic inflammation |
3 (1.5) |
Benign neoplasms (4.5) |
Hyperplastic polyp |
6 (3.0) |
Fundic gland polyp |
2 (1.0) |
Tubular adenoma |
1 (0.5) |
Premalignant (1.5) |
Intestinal metaplasia/dysplasia |
2 (1.0) |
Barrett’s oesophagus with dysplasia |
1 (0.5) |
Malignant (24.0) |
Gastric adenocarcinoma |
31 (15.5) |
Oesophageal squamous cell carcinoma (SCC) |
15 (7.5) |
Carcinoma with neuroendocrine differentiation |
1 (0.5) |
Gastrointestinal stromal tumour (GIST) |
1 (0.5) |
Table 2. Distribution of histopathological diagnoses (n = 200)
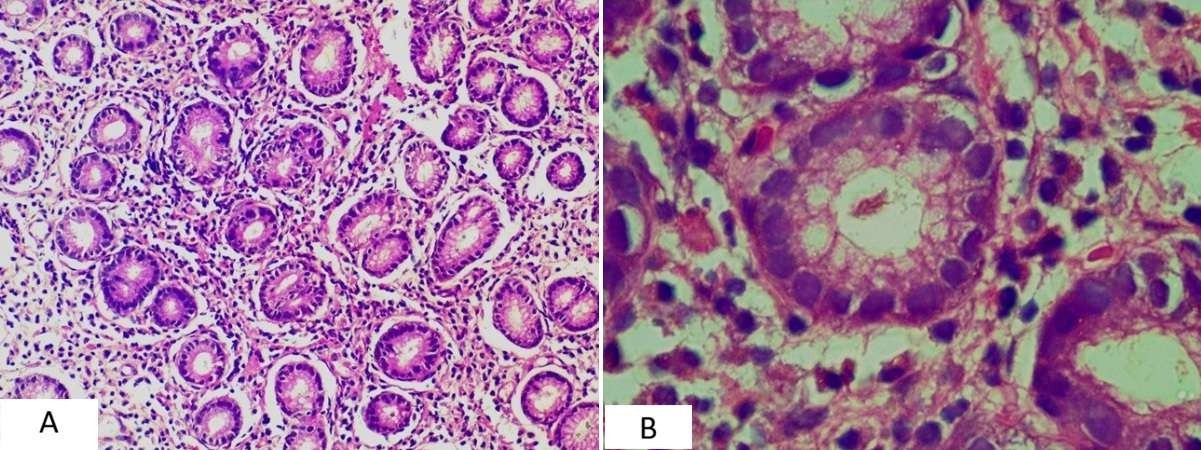
Figure 1. (A – B). Histological features of inflammatory lesions. (A) Chronic gastritis showing dense mononuclear infiltrates in the lamina propria (H&E, 200×). (B) Chronic gastritis with Helicobacter pylori organisms adherent to the gastric epithelium (H&E, oil immersion).
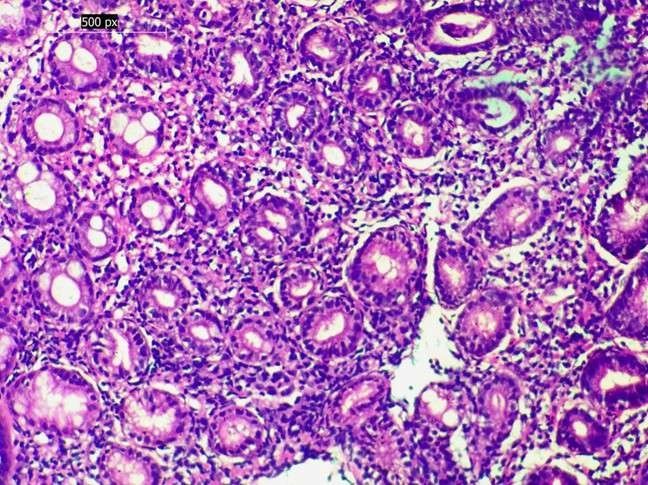
Figure 2. Premalignant change in chronic gastritis. Chronic gastritis with intestinal metaplasia, characterised by replacement of gastric epithelium by intestinal-type columnar cells with goblet cells (H&E, 400×)
Helicobacter pylori were identified in 60 cases (30%) among patients with chronic gastritis. Metaplasia of the intestine was seen in 14 cases (7%) and neutrophilic activity was seen in 23 cases (11.5%). Less common were features of atrophy (1%), parietal-cell hyperplasia (2.5%), and dysplasia (1.5%). These histological changes were all related to chronic gastritis only (see Table 3).
Feature |
Associated pathology |
n (%) |
Helicobacter pylori |
Present in chronic gastritis |
60 (30.0) |
Intestinal metaplasia |
Chronic gastritis |
14 (7.0) |
Neutrophilic activity |
Chronic gastritis |
23 (11.5) |
Atrophy |
Chronic gastritis |
2 (1.0) |
Parietal cell hyperplasia |
Chronic gastritis |
5 (2.5) |
Dysplasia |
Chronic gastritis |
3 (1.5) |
Abdominal pain |
Chronic gastritis (pylorus) |
Inflammatory > neoplastic |
Dyspepsia |
Chronic gastritis (pylorus) |
Often non-specific |
Dysphagia |
Oesophageal SCC / GEJ adeno |
Strong malignant association |
Reflux symptoms |
Chronic gastritis / Barrett’s |
Premalignant changes rare |
Ulceroproliferative growth |
Adenocarcinoma / SCC |
High predictive value |
Antral/periantral gastritis |
Chronic gastritis |
Strong correlation |
Abdominal pain |
Chronic gastritis (pylorus) |
Inflammatory > neoplastic |
Dyspepsia |
Chronic gastritis (pylorus) |
Often non-specific |
Table 3. Additional histological features (n = 200)
The most common cases of chronic gastritis were found in patients who presented with abdominal pain and dyspepsia, and related in many cases to biopsies that were taken in the pylorus. A strong relationship was shown between malignant lesions and older age; the lesions were observed in patients over 40 years old and with a strongest male predominance in the population 3: 1). Ulcer proliferative growths had nearly a perfect correlation with malignant histology by endoscopy, whereas antral/perinatal gastritis was associated with inflammatory lesions (see Table 3, Figures 3-5).
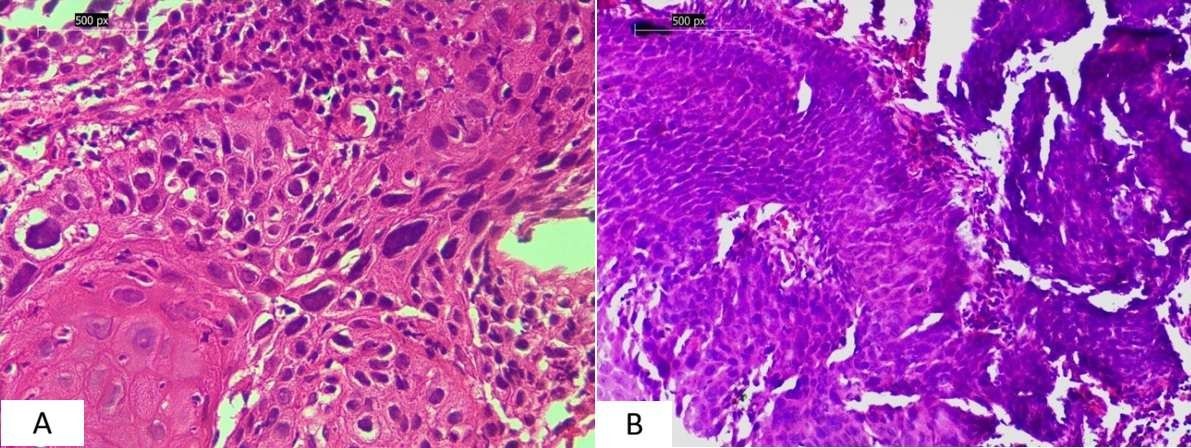
Figure 3. (A – B). Oesophageal squamous cell carcinoma. (A) Well-differentiated squamous cell carcinoma with keratin pearl formation (H&E, 400×). (B) Poorly differentiated squamous cell carcinoma showing marked pleomorphism and high mitotic activity (H&E, 200×)
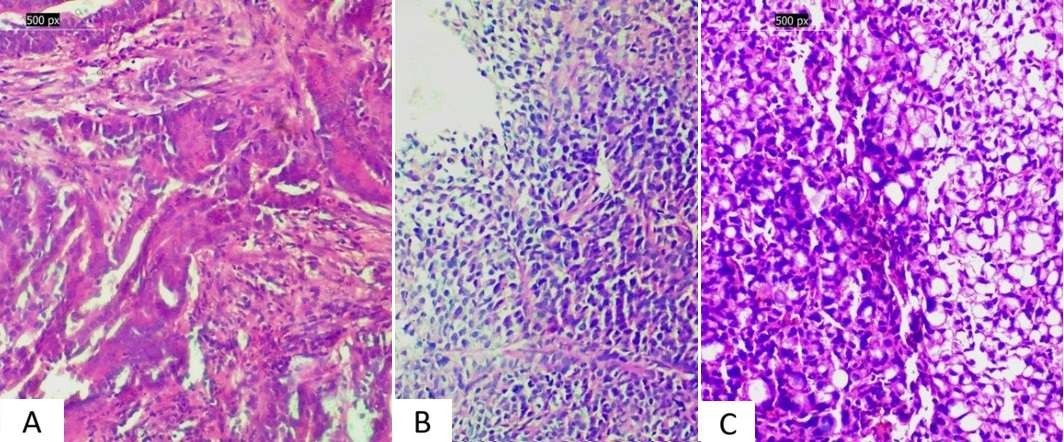
Figure 4. Gastric adenocarcinoma spectrum. (A) Gastric adenocarcinoma showing glandular architecture (H&E, 200×) (B) Poorly cohesive adenocarcinoma with loss of gland formation (H&E, 200×) (C) Signet ring cell carcinoma characterised by intracytoplasmic mucin displacing the nucleus (H&E, 200×)
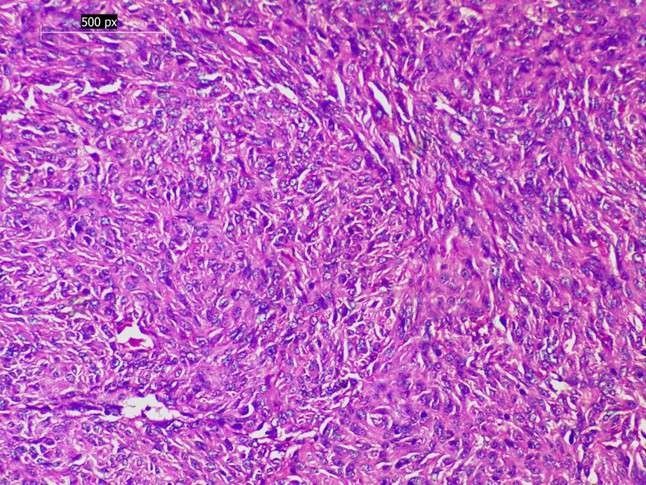
Figure 5. Gastrointestinal stromal tumor (GIST). Proliferation of spindle-shaped cells arranged in fascicles, consistent with GIST (H&E, 200×)
Discussion
This study was aimed at clarifying the range of histopathological lesions that is most commonly detected in upper gastrointestinal biopsies in a tertiary care centre in Kerala and to determine how the diagnoses in these cases are reflected by the endoscopic observations and the clinical presentation of the patients. The statistics indicate that chronic gastritis was the dominant element of the inflammatory spectrum after which malignant neoplasms (mainly gastric adenocarcinomas and oesophageal squamous cell carcinomas) constituted about a quarter of all the cases. Premalignant changes like areas of metaplasia of the intestines and Barretts oesophagus with dysplasia were not common but clinically important. Most of the patients were above 40 years in age, there was a slight dominance of males and the most common presenting complaint was abdominal pain. Endoscopic analysis showed that antral gastritis was predominant and pyloric region was the most frequent location of biopsy. H. pylori were also found to be a positive etiologic agent in gastric pathology, with approximately a third of chronic gastritis cases being positive.
The cohort was mainly composed of persons over 40 years (80.5%), but the preponderance was slightly more male. These populations agree with other recent Indian research findings that also demonstrate that there is a greater occurrence of upper GI pathology in middle-aged and elderly adults and that there is a male excess in both inflammatory and malignant lesions [2,16,30]. The commonest symptom was abdominal pain, which was then followed by dyspepsia and dysphagia as in Rana et al. and Gunay et al. [1,18]. These observations reiterate the fact that nonspecific symptoms often cloud out major pathology and hence the importance of biopsy in symptomatic patients, especially above 40 years.
The most typical biopsy site was the pylorus (70.5%), as the percentage of patients who reported abdominal pain and dyspeptic symptoms was high. Antral gastritis was the most frequent endoscopic finding with almost half the patients having this condition. Other Indian series show better proportions of oesophageal biopsies [17,20]. These regional variations would probably be variations in patterns of referrals, diet, and exposure to risk factors.
Histopathological diagnosis revealed inflammatory lesions (70%), and chronic gastritis was the most common subtype (97.8%). This trend is consistent with the findings of other Indian cohorts and studies in the region where gastritis is the most common biopsy result [2,19]. H. pylori have been detected in about a third of chronic gastritis and is similar to South Asian studies, but a bit lower than global estimates which frequently exceed 50% [21,26]. The regional differences in prevalence might reflect the advances in sanitation, nutrition, exposure to antibiotics, and the diagnostic techniques. Notably, other features (intestinal metaplasia, neutrophilic activity, and atrophy) were also detected, thus highlighting their contribution to the cascade of Correa towards gastric carcinogenesis [27,28].
The proportion of benign lesions was 4.5% of biopsies, and all were presented in the form of polyps or nodules on endoscopy. Hyperplastic polyps had the highest rate then fundic gland polyps and tubular adenomas as per the distribution by Jonnalagadda, et al. [16] and Rani, et al. [15] Premalignancies were infrequent (1.5%), but seemed to have clinical importance, such as Barrett oesophagus with dysplasia, and chronic gastritis with underlying foci of dysplasia. This proportion is lower than some studies conducted in India and the West, but lack of systematic sampling as per the Sydney protocol might have led to under-detection [11,12]. With the known progressive development of intestinal metaplasia and Barrett carcinoma oesophagus, diagnosis of the lesions should be closely followed.
At least 24% of the cases were diagnosed with malignancies, mostly in patients who were of age over 40 years, and those who were males. The most common neoplasm was gastric adenocarcinoma, which is followed by oesophageal squamous cell carcinoma and gastroesophageal junction adenocarcinoma. This distribution is regional and epidemiology, as gastric adenocarcinoma is still very common and SCC is dominant in the oesophageal cancer [31,32]. The histological subtyping showed that the majority of gastric cancers were adenocarcinomas, including poorly cohesive carcinomas with signet ring morphology, which was not the first time that consonant results were obtained in India [2,19]. The fact that all the oesophageal SCCs were presented as ulceroproliferative lesions, with the majority being moderately differentiated, is consistent with those by Jonnalagadda, et al. [16], and Rauta, et al. [17]. These correlations between endoscopic appearance and histologic subtype can be used to give useful hints at early suspicion and directed biopsy.
The finding that H. pylori has remained relevant in gastric pathology and that eradication therapy has a role to play in reducing long-term risks of ulceration and malignancy is supported by the fact that it has been identified in a significant proportion of cases. In addition to this, the premalignant changes though rare in occurrence have been detected which underscores the need to have systematic biopsy procedures and follow-up measures which will enable timely intervention. To clinicians in Kerala and similar environments, the findings recommend caution in the assessment of dyspeptic patients especially those who are above 40 and those who exhibit persistent or numerous symptoms.
This study has some strengths such as its real-life design, consecutive sampling and systematic correlation of histopathology, clinical presentation and endoscopic information. Weaknesses include small sample size compared to the overall number of biopsies done within the institution, non-systematic sampling in terms of the Updated Sydney System, and no follow-up of premalignant lesions. The study is a single-centre study, and the results might not be applicable to the wider population.
Future studies would include multicentric cohorts, increase sample sizes and standardised biopsy procedures in order to improve the detection of premalignant conditions. It is recommended that longitudinal follow-up of intestinal metaplasia patients or those with Barretts oesophagus be done in an attempt to gain a better understanding of the progression to malignancy. Moreover, the risk of the disease in various regions could be further explained by molecular investigations of H. pylori strains and host susceptibility factors.
Conclusion
Finally, the histopathology of upper GI biopsies in this Kerala cohort is almost entirely dominated by inflammatory lesions, especially chronic gastritis, but malignant lesions represent a significant burden. Although they are less prevalent, benign and premalignant lesions are still clinically significant with regard to cancer prevention. These results highlight the importance of histopathology as being an indispensable component of clinical and endoscopic assessment, early detection of neoplastic change, and a method of determining the correct approach to patient management when the patient presents with upper GI symptoms.
Acknowledgement
The authors sincerely acknowledge the support and cooperation of institutional departments and technical personnel involved in the conduct of this study. The authors also thank the patients whose biopsy samples formed the basis of this study.
Conflicts of interest
The authors declare that there are no conflicts of interest regarding the publication of this manuscript.
References
- Rana M, Nahar K, Nahar N, Ahmed M, Salam T, et al. (2023) Clinical indication of upper gastrointestinal endoscopy and its outcome. Saudi J Med Pharm Sci 9: 24-28.
- Naik P, Das M (2023) A study of histopathological features of upper gastrointestinal tract endoscopic biopsies in a tertiary care centre. Panacea J Med Sci 13: 493-496.
- Valdez LM, Gotuzzo E, Thielman NM, DuPont HL (2011) Gastrointestinal symptoms. In: Elsevier eBooks 916–924.
- Cook GC (2009) Tropical gastroenterological problems. In: Elsevier eBooks 107–141.
- Campbell IS, Howell JD, Evans HH (2016) Visceral vistas: Basil Hirschowitz and the birth of fiberoptic endoscopy. Ann Intern Med 165: 214-218. [Crossref]
- Ahlawat R, Hoilat GJ, Ross AB (2023) Esophagogastroduodenoscopy. StatPearls - NCBI Bookshelf. Aug 8. [Crossref]
- Pouw RE, Barret M, Biermann K, Bisschops R, Czakó L, et al. (2021) Endoscopic tissue sampling–Part 1: Upper gastrointestinal and hepatopancreatobiliary tracts. European Society of gastrointestinal endoscopy (ESGE) guideline. Endoscopy 53: 1174-1188. [Crossref]
- Loughrey MB, Shepherd NA (2021) The indications for biopsy in routine upper gastrointestinal endoscopy. Histopathology 78: 215-227. [Crossref]
- Alberto B, Chiara M, Alessandra V, Ginevra C, Antonio N, et al. (2018) A non-invasive method for the diagnosis of upper GI diseases. Acta Biomed 89: 40-43. [Crossref]
- Dubcenco E, Jeejeebhoy KN, Petroniene R, Tang SJ, Zalev AH, et al. (2005) Capsule endoscopy findings in patients with established and suspected small-bowel Crohn's disease: Correlation with radiologic, endoscopic, and histologic findings. Gastrointest Endosc 62: 538-544. [Crossref]
- Eshmuratov A, Nah JC, Kim N, Lee HS, Lee HE, et al. (2010) The correlation of endoscopic and histological diagnosis of gastric atrophy. Dig Dis Sci 55: 1364-1375. [Crossref]
- Lim JH, Kim N, Lee HS, Choe G, Jo SY, et al. (2013) Correlation between endoscopic and histological diagnoses of gastric intestinal metaplasia. Gut Liver 7: 41. [Crossref]
- Poudel A, Regmi S, Poudel S, Joshi P (2013) Correlation between endoscopic and histopathological findings in gastric lesions. J Universal Coll Med Sci 1: 37-41.
- Sharma S, Makaju R, Dhakal R, Purbey B, Gurung RB, et al. (2015) Correlation between endoscopic and histopathological findings in gastric lesions. Kathmandu Univ Med J 13: 216-219. [Crossref]
- Rani D, Bhuvan S, Gupta A (2019) A study of morphological spectrum of upper gastrointestinal tract lesions by endoscopy and correlation between endoscopic and histopathological findings. Indian J Pathol Oncol 6: 28-34.
- Jonnalagadda K, Karre S, Thungaturthi SR, Gorrela VD (2019) Histopathological spectrum of upper gastrointestinal endoscopic biopsies. Indian J Pathol Oncol 6: 422-427. [Crossref]
- Rauta S, Baisakh P, Sahoo AK, Panda DK, Baisakh MR, et al. (2024) Correlation of endoscopic and histopathological diagnoses in upper gastrointestinal tract lesions: A cross-sectional study. Cureus 16: e69553. [Crossref]
- Günay E, Özkan E, Odabaşı HM, Abuoğlu HH, Eriş C, et al. (2013) Symptom-histopathology relation in upper GI endoscopy. Ulus Cerrahi Derg 29: 115-158. [Crossref]
- Bhide S, Lahane R (2024) Histopathological spectrum of upper gastrointestinal endoscopic biopsies in a rural teaching hospital. Med Lab J 18: 1–3.
- Koirala S, Khadka A, Bhusal S, Shrestha R, Prasai A, et al. (2024) Histopathological spectrum of the upper gastrointestinal tract endoscopic biopsies at a tertiary hospital: A descriptive cross-sectional study. J Nepal Med Assoc 62: 453-457. [Crossref]
- Cortez Nunes F, Mateus TL, Aguieiras C, Louro R, Peixe B, et al. (2025) Prevalence and diagnostic comparison of helicobacter pylori and non-helicobacter pylori helicobacter infections in patients undergoing upper gastrointestinal endoscopy with gastric biopsy in Algarve, Portugal. Microorganisms 13: 1684. [Crossref]
- Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, et al. (2007) The strengthening the reporting of observational studies in epidemiology (STROBE) statement: Guidelines for reporting observational studies. The Lancet 370:1453-1457.
- Veerendrasagar RS, Nandish VS (2020) Histopathological spectrum of lesions of upper gastrointestinal tract endoscopic biopsies. Intl J Clin Diagnos Pathol 3: 39–42.
- Bancroft JD, Gamble M (2008) Theory and practice of histological techniques. Elsevier Health Sci.
- El-Zimaity HM (2000) Accurate diagnosis of Helicobacter pylori with biopsy. Gastroenterol Clin North Am 29: 863-869. [Crossref]
- Chey WD, Howden CW, Moss SF, Morgan DR, Greer KB, et al. (2024) ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am J Gastroenterol 119: 1730–1753. [Crossref]
- Correa P, Piazuelo MB (2011) The gastric precancerous cascade. J Dig Dis 13: 2–9. [Crossref]
- Correa P, Piazuelo MB (2008) Natural history of Helicobacter pylori infection. Dig Liver Dis 40: 490–496. [Crossref]
- Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology 76: 182–188. [Crossref]
- Venkatesh V, Thaj RR (2019) Histopathological spectrum of lesions in gastrointestinal endoscopic biopsies: A retrospective study in a tertiary care center.
- Bai Z, Wang H, Shen C, An J, Yang Z, et al. (2024) The global, regional, and national patterns of change in the burden of nonmalignant upper gastrointestinal diseases from 1990 to 2019 and the forecast for the next decade. Int J Surg 111: 80–92 [Crossref]
- Saydam ŞS, Molnar M, Vora P (2023) The global epidemiology of upper and lower gastrointestinal bleeding in general population: A systematic review. World J Gastrointest Surg 15: 723–739. [Crossref]





