Abstract
Background
The diagnosis of acute myocardial infarction (AMI) is currently confirmed by the determination of plasma Troponin I (pTi). Based on the previous reports, that the platelets from AMI subjects aggregated by different aggregating agents could not be inhibited by acetyl salicylic acid (aspirin), the feasibility of the diagnosis of AMI was studied.
Methods
Subjects between 40-60 years with either AMI (n=50, M=35, F=15) or ACS (n=30, M=20, F=10) and an equal number of age and sex matched normal volunteers participated in the study. The inhibition of 80 µM aspirin of platelet aggregation induced by 2.0 µM ADP was studied by incubating the platelet rich plasma (PRP) with the compound for 30 min at 37°C and aggregation was performed in an optical aggregometer.
Results
While in the case of normal PRP, aspirin induced inhibition was ≈100%. In the case of ACS, the percent inhibition was 63.2% compared to the cases of AMI where no inhibition (i.e. 0% inhibition, p<0.001) by aspirin in ADP induced platelet aggregation was found (sensitivity= 92% and specificity = 100%).
Conclusion
It was feasible to diagnose the occurrence of AMI by the absence of the inhibitory effect of aspirin on platelet aggregation.
Key words
platelet aggregation, myocardial infarction diagnosis, troponin, aspirin.
Introduction
The aggregation of platelets by different platelet aggregating agents including ADP, l-epinephrine, collagen or thrombin is known to be essential for the lifesaving process of blood coagulation [1]. On the other hand, excessive platelet aggregation on the site of atherosclerotic plaque rupture on the wall of the coronary artery is reported to result in the development of thrombus, a micro-aggregate of platelets, which may sometimes lead to the blockade of the normal blood circulation in the musculature of the heart, and that in consequence may result in acute coronary syndrome (ACS) [2]. Although the aggregation of platelets on the arterial plaque rupture site may develop into ACS, the aggregation of platelets, sometime may also block the pericardial artery that might result in the cardiac cells death which appeared as dark patchy areas of infarcts developed due to the death of heart cells that ultimately could lead to acute myocardial infarction (AMI), which when massive, could result in the death of the victims [3]. The transmural infarcts depending on the extent of damage in the myocardium that may or may not be always expressed as abnormal Q wave but may also be involved in the appearance of abnormal ST and T waves in 50% of the cases in the ECG resulting in St-Elevated Myocardial Infarction (STEMI) or non-STEMI, and as such, the ECG may not be diagnostically useful to determine the occurrence of AMI. Although the chest pain is usually severe and lasts longer than that in the case of ACS, the pain itself is known to be subjective in nature and may be a simulated event other than by the AMI itself. And, as such, the diagnosis of AMI is usually confirmed by the determination of the plasma troponin I (pTi) by Enzyme Linked Immuno sorbent Assay (ELISA) using troponin I antibody.
However, both troponin I (Ti) and its antibody are expensive and furthermore, the ELISA itself may not always be adequately fast enough to confirm the occurrence of AMI in demanding situation which may be needed for the possible therapeutic intervention for the condition. For these reasons, the laboratory diagnosis of AMI is not available, not necessarily by choice, in many parts of the world.
We have recently reported that the Platelet Rich Plasma (PRP) from AMI patients pre-incubated with 80 µM acetyl salicylic acid (aspirin), for 30 min at 37°C failed to inhibit aggregation of platelets initiated by different platelet aggregating agents including ADP when compared to that in the normal PRP or in the PRP from ACS. Based on these preliminary studies we have studied the possibility of confirming the occurrence of AMI by optical aggregometry even in the absence of the determination of the pTi.
We herein report the feasibility of confirming the occurrence of AMI differentiated from the occurrence of ACS by determining the aspirin induced inhibition of platelet aggregation induced by ADP using an aggregometer. The occurrence of AMI was confirmed by parallel and simultaneous determination of the pTi by ELISA.
Methods and materials
Ethical clearance
The research project, ‘‘Laboratory diagnosis of acute myocardial infarction by optical aggregometry of platelets’’ required nominal amount of blood (2 mL) from the patients with AMI or ACS and from the normal volunteers. The Institutional Review Board, Human & Animal Research Ethics Committee, Sinha Institute Of Medical Science And Technology, Kolkata, India approved the study on the condition that followed the approved Human Ethics Protocol strictly in accordance with 1964 Helsinki declaration and no deviation in the study was allowed without the prior written permission of the board. All the participants were between 40-60 years old. No mentally retarded, pregnant women or prisoner took part in the study. All the volunteers signed an informed consent form prior to their participation in the study. In the cases of ACS or AMI subjects, permission to participate in the study was also obtained from the kin of the patient by signing a judicial affidavit. The attending physicians decided whether the blood sample (2 ml) might be withdrawn from the patient without putting the subject in any danger. The blood was withdrawn by the attending physician or by a licensed nurse. It was ensured that the volunteers had no other life threatening infection. The committee inspected the progress and problems of the current investigation routinely.
Chemicals
The goat anti-rabbit immunoglobulin G-alkaline phosphatase and ADP were obtained from Sigma Aldrich. Enzyme Linked Immunosorbent Assay (ELISA) maxisorp plates were from Nunc, Rosaklide, Denmark. Troponin I, Troponin I antibody, CK-MB, CK-MB antibody were obtained from Abcam. Acetyl Salicylic Acid (aspirin) was obtained from Medica Zydus Healthcare. All other chemicals used were of analytical grade.
Selection of AMI patients
A total number of 50 patients (n=50, M=35, F=15, between the ages of 40-60 years with chest pain that lasted >120h participated in the study. These subjects were admitted to the Intensive Care Unit in the Calcutta Medical College and Hospital. The patients with diabetes mellitus or life threatening infection were not included in the study. As paricarditis, pulmonary embolism, aortic and intestinal disorders are known to stimulate chest pain due to AMI or ACS, patients with these conditions were carefully excluded from the study. All participants who have been hospitalized with any condition within the last six months or patients who have received aspirin within 2 weeks were excluded from the study.
Selection of normal volunteers
Equal number of age and sex matched normal volunteers were also included in the study. None of the normal volunteers had diabetes mellitus or have any history of systemic hypertension at presentation. None of the normal volunteers had any life threatening infection or had been hospitalized for any condition within the last six months. None of the female volunteers had ever received any contraceptive medications. None of the volunteers had taken any aspirin or any cardiac medication within the last 4 weeks.
Diagnosis of AMI
All AMI patients were admitted in the intensive cardiac care unit in the hospital with the characteristic chest pain as described above. AMI patients with both STEMI and non-STEMI were included in the study. All selected patients had developing Q waves in ECG. In those cases, where the occurrence of Q waves in the ECG could not be clearly demonstrated clearly were excluded from the study. The occurrence of AMI was confirmed by the pTi levels by ELISA.
Diagnosis of ACS
As a part of the intended study, 30 patients (n=30, M=20, F=20) between the ages of 40-60 years were included in the study. These patients had characteristic chest pain. Those patients who had bundle branch block and the regions of cardiac ischemia could not be ascertained by ECG were excluded from the study.
The occurrence of ACS was confirmed by the determination of CK-MB in the blood sample by withdrawing the sample within 6h of admission in the hospital.
The blood samples either in the case of AMI or ACS were obtained from the subjects before the initiation of any cardiac therapy to minimize ambiguity.
Study protocol
The selected patients were randomized and an arbitrary number was assigned to the each blood sample from AMI, ACS and from normal volunteers and kept under the supervision of a clerical staff who was not an investigator. The investigator had no knowledge whether the withdrawn blood samples was from an AMI or ACS or from a normal volunteer. Only after the completion of the study, the “key” to the information regarding the condition of the participant was available to the investigators for statistical analyses. All blood samples were studied as soon as practicable after the withdrawal of the sample.
Preparation of platelet rich plasma (PRP)
The PRP from the subjects was prepared by centrifugation as described before [4].
The platelet free plasma (PFP) used for the determination of CK-MB or pTi was prepared by centrifuging the blood samples at 10,000 g at 0°C [4].
2021 Copyright OAT. All rights reserv
Inhibition of platelet aggregation by aspirin
Typically, the PRP from the subjects with AMI, ACS or from the normal volunteers was incubated with 80 µM aspirin for 30 min at 37°C. The aggregation of platelets was initiated by treating the PRP with 2 µM ADP pre-incubated with or without aspirin [4]. In control experiments, the PRP was treated with an equal volume of 0.9% NaCl incubated at 37°C for 30 min.
Preparation of aspirin solution
Freshly prepared solution of aspirin was made by dissolving aspirin in de-ionized water and the pH was maintained at 7.0 by adding 0.1M NaHCO3. The aspirin solution was discarded after use.
Assay of plasma Troponin I and CK-MB
The pTi and CK-MB levels were determined by ELISA by using Troponin I antibody and CK-MB antibody.
Statistical analysis
The obtained results are presented as +/- standard deviation, while the significance of the results was determined by using Student’s t-test. Values of p<0.05 were considered significant. Where appropriate, the Pearson´s correlation coefficients ® of the results were also determined. Pearson score “r”, is such that -1 ≤ r ≤ +1 is acceptable where the (+) and (-) signs are designated as positive linear correlations and negative linear correlations, respectively. Graph Pad Prism software (Graph Pad Software, San Diego, and USA) and Micro Cal origin 6.0 software were used for the statistical analyses. The diagnostic test evaluation and Receiver Operator Curve (ROC) analysis was performed using statistical software, MedCalc, Belgium. According to ROC analysis a test with perfect discrimination (no overlap in two distributions) has a ROC curve that passes through the upper left corner (100% sensitivity, 100% specificity). Therefore, closer the ROC curve is to the upper left corner, the higher is the overall accuracy of the test [5]. The area under curve (AUC) was quantitated which is a measure of how well a parameter can distinguish between two diagnostic groups (diseased/normal).
Results
The effect of aspirin on the inhibition of platelet aggregation induced by ADP in PRP from the subjects with ACS, AMI and from the normal volunteers
When the PRP from these subjects were incubated with 80 µM aspirin for 30 min at 37°C and the platelet aggregation was initiated by treating the PRP with 2.0µM ADP, it was found that only in the case of PRP from AMI subjects, the compound failed to inhibit (0% inhibition) platelet aggregation. In contrast, in the case of ACS (percent inhibition ranging from 52%-68%) or in the case of normal PRP the platelets were inhibited by 100% (Figure 1, Panel A). The receiver operator curve (ROC) analysis showed 92% sensitivity (95% confidence interval (CI): 91.0-100.0) and 100% specificity (95% CI: 91.0-100.0, P<0.0001). The area under curve (AUC) as tested by ROC analysis came to be equal to 0.952 (Figure 1, Panel B).
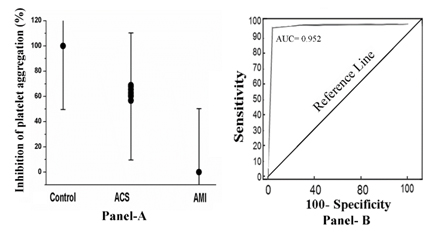
Figure 1. The percent inhibition of platelet aggregation and receiver operator curve analysis in PRP by aspirin in the case of normal, ACS and AMI subjects. Panel A: The PRP from normal, ACS and AMI subjects were prepared as described in Materials and Methods and incubated with 80 µM aspirin for 30 min at 37°C. After incubation, platelet aggregation was initiated by adding 2 µM ADP and studied in an optical aggregometer. Panel B: ROC curve analysis of the inhibition of platelet aggregation in which sensitivity is plotted against specificity for different cut-off points. Each point on the ROC curve represents a sensitivity/specificity pair corresponding to a particular decision threshold as described in Methods and Materials.
Relation between the pTi and aspirin induced inhibition of platelet aggregation by ADP in AMI, ACS and in normal PRP.
As shown in (Figure 2, Panel A), the increase of pTi was found to be inversely related to the extent of aspirin induced inhibition of platelet aggregation. In other words, the increase of pTi which was maximal in the case of AMI also led to the development of maximal resistance of the platelets to the inhibitory effect of aspirin (Co-efficient of correlation, Pearson’s r= -0.967, with p<0.001), and the aspirin induced inhibition of the platelet aggregation was maximally related to the total absence of pTi in normal and the pTi level in ACS was more than that in normal plasma that demostrated troponin I was also present in ACS plasma albeit less than that in AMI. The receiver operator curve (ROC analysis) showed 85% sensitivity (95% confidence interval (CI): 81.0-92.0) and 78% specificity (95% CI: 75.0-80.0, P<0.0001). The area under curve (AUC) as tested by ROC analysis came to be equal to 0.739 (Figure 2, Panel B).
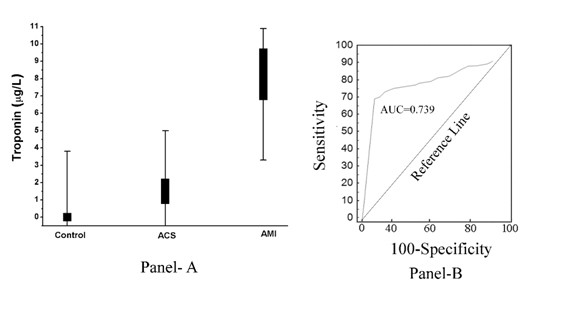
Figure 2. Plasma Troponin I level in normal, ACS and AMI subjects and its ROC analysis. Panel A: The pTi level was determined by ELISA using Troponin I antibody as described in Materials and Methods. Each point is actually a overlapping of 50 normal, 30 ACS and 50 AMI patients. Panel B: ROC curve analysis of the pTi levels in normal (non-diseased), AMI (diseased) patients was constructed using MedCalc as described in Methods and Materials.
Discussion
The acute myocardial infarction, reported to be cause by thrombosis that results in cardiac cell death could arguably be the most dangerous thrombotic disorder that leads to large number of death among the victims [3]. In contrast to ACS, where the recurrence of the condition is reported to be prevented by aspirin through the inhibition of excessive aggregation of platelets, the platelets in AMI are reported to be resistant to the inhibitory effect of the compound due to the appearance of dermcidin isoform-2 (DCN-2) in the circulation [6]. Unfortunately, due to the lack of aspirin induced inhibition of platelet aggregation, most of the first time victims had a recurrence of AMI within months and succumb to the condition. As DCN-2 was reported to be a potent inhibitor of aspirin induced nitric oxide (NO) synthesis [7]. It has been recently reported that by removing the bound DCN-2 from the platelet surface through increased NO synthesis in platelets, it is possible to resensitize the AMI platelets to the aspirin effect [7].
Not only the treatment and prevention of AMI through appropriate therapeutic intervention remains problematic, even the laboratory diagnosis of the condition, differentiated from the occurrence of ACS, remains prohibitively expensive for many laboratories in the world which is usually needed for the confirmation of AMI by the pTi level. This is due to the fact that both troponin I and its antibody are not only expensive, but the assay itself by ELISA needs special equipment and necessary chemicals and supplies which could be a time consuming procedure and in needful situation where a rapid and confirmatory occurrence of AMI is essential for the therapeutic intervention. Furthermore, the interaction between an antigen and its antibody does not follow the well-known chemical stoichiometry, and, as such, the assay itself may present problems in the validity of the interpretation of the data. Also, the diagnosis of ACS, differentiated from that of AMI, by the determination of plasma CK-MB by itself is an expensive procedure.
The results presented above strongly suggested the feasibility of the diagnosis of AMI, differentiated from the occurrence of ACS, by simple platelet aggregometry using ordinary laboratory aggregometer within an hour (Sensitivity= 92% and Specificity= 100% as determined by ROC analysis). The curve of the diagnostic test as described in materials and methods passes close through the upper left corner (Figure 1, Panel B) implying close to 100% accuracy of the diagnostic test compared to pTi (Figure 2, Panel B).
Also as the aggregation of platelets in PRP cannot be inhibited by aspirin in AMI, but the platelets from the PRP from ACS subjects could be, the diagnosis of AMI by platelet aggregometry could be an economic as well as “all or nothing” event i.e. the aggregation of AMI platelets could or could not be achieved at all by the treatment of PRP with aspirin, in contrast the aggregation of platelets from the ACS patients could be inhibited markedly by the same compound (Figure 1). However, it was found that even the platelets from ACS were somewhat more resistant to the inhibitory effect of aspirin than that in the case of normal platelets. On the other hand, the platelets from AMI was maximally resistant to the effect of aspirin that was inversely related to the pTi levels (Coefficient of correlation (r) between the pTi level and % of inhibition by aspirin was -0.967, p<0.001).
It should be mentioned in the above context that a rapid and confirmatory diagnosis of AMI is necessary for the use of thrombolytic therapy which may reduce the mortality rate in AMI by 30-50% [8]. It should also be noted that about 50% of the patients with increased pTi do not have either ST elevation or Q waves in ECG, and as such, the platelet aggregometric diagnosis of AMI could be an even diagnostically more useful method than ECG, and at the same time it is much less expensive than the ELISA for pTi determination.
The quantitation of pTi in the circulation of AMI patients is usually carried out as a confirmatory marker protein for AMI. However, in a preliminary study it was found that pTi was also an inhibitor of NO synthesis and a potent inducer of platelet aggregation in the ranges (nM) similar to the levels of pTi in the AMI plasma (unpublished). However as the plasma level of troponin I in ACS was more than that in normal plasma, but much less than that in case of AMI, these results suggested that the increase of pTi did occur also in ACS but almost 6 times less than that in AMI. These results nevertheless suggested that the increased pTi compared to normal plasma alone may not necessarily confirm the occurrence of AMI differentiated from the possible occurrence of ACS.
We have recently reported that the characteristic anginal pain in AMI was related to the severe reduction of NO level in plasma that was due to the inhibition of NO synthesis by DCN-2 or other systemic inhibitors of nitric oxide synthase [9]. As pTi was found to be a novel platelet aggregating agent (unpublished), through the inhibition of NOS in platelets, it is possible that pTi itself could be involved in the anginal pain, atleast partly, in the AMI. The spillage of pTi in the circulation from the cardiac muscles could be thrombogenic by itself and may contribute to the development of resistance to the inhibitory effect of aspirin in AMI platelets similar to the effect of DCN- 2 resulting in the initiation or to the aggravation of the condition. And thus, the presence of pTi may itself could have an ominous role in the development of resistance to the inhibitory effect of aspirin in the platelets in AMI leading to the recurrence of the condition.
References
- Colman RW, Walsh PN (1987) Haemostasis andthrombosis. In: Colman RW, Hirsh J, Maeder VJ, Salzman EW, eds. Philadelphia, PA (USA), J.B. Lippincott: 594-605.
- Fuster V, Badimon J, Chesebro JH, Fallon JT (1996) Plaque rupture, thrombosis, and therapeutic implications. Haemostasis 4: 269-284. [Crossref]
- Page DL, Caulfield JB, Kastor JA, DeSanctis RW, Sanders CA (1971) Myocardial changes associated with cardiogenic shock. N Engl J Med 285: 133-137. [Crossref]
- Chakraborty K, Khan GA, Banerjee P, Ray U, Sinha AK (2003) Inhibition of human blood platelet aggregation and the stimulation of nitric oxide synthesis by aspirin. Platelets 14: 421-427. [Crossref]
- Zweig MH1, Campbell G (1993) Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem 39: 561-577. [Crossref]
- Bank S, Jana P, Maiti S, Guha S, Sinha AK (2014) Dermcidin isoform-2 induced nullification of the effect of acetyl salicylic acid in platelet aggregation in acute myocardial infarction. Sci Rep 4: 5804. [Crossref]
- Ghosh R, Karmohapatra SK, Bhattacharya M, Bhattacharya R, Bhattacharya G (2011) The appearance of dermcidin isoform 2, a novel platelet aggregating agent in the circulation in acute myocardial infarction that inhibits insulin synthesis and the restoration by acetyl salicylic acid of its effects. J Thromb Thrombolysis 31: 13-21.
- Moreno FL, Villanueva T, Karagounis LA, Anderson JL (1994) Reduction in QT interval dispersion by successful thrombolytic therapy in acute myocardial infarction. TEAM-2 Study Investigators. Circulation 90: 94-100. [Crossref]
- Ghosh R, Ray U, Jana P, Bhattacharya R, Banerjee D, et al. (2014) Reduction of death rate due to acute myocardial infarction in subjects with cancers through systemic restoration of impaired nitric oxide. PLoS One 9: e88639. [Crossref]


