Abstract
A simple microreactor was fabricated by vertically-aligned multi-walled carbon nanotubes (MWCNTs) on the mc-Si substrate. MWCNTs were synthesized by home-made equipment aided with chemical vapor deposition (CVD). The wettability of the surfaces of microreactor with the synthesized MWCNTs, Au coating and plasma irradiation were investigated, respectively. Results show that the initial surface with the MWCNTs film behaves as a super-hydrophobic surface. However, it changes to a super-hydrophilic surface after coated with Au or irradiated by plasma. It shows that microreactor with treated MWCNTs film can be used as potential applications for bioanalysis.
Keywords
Multi-walled carbon nanotubes (MWCNTs); Chemical vapor deposition (CVD), Bioanalysis microreactor; Wettability; Hydrophobic; Hydrophilic; Lab-on-a-chip (LOC); μTAS.
Introduction
Nanomaterials are playing the more and more important role in electronic, biological, chemical, and medical applications. In particular, carbon nanotubes (CNTs) have been attracting significant attention because of their unique characteristics [1]. CNT electrodes and CNT-based reactors have been applied as electrodes in fuel cells [2, 3], and reaction fields in biosensors [4, 5], etc.
The development of lab-on-a-chip (LOC) devices, a term for “micro-total analysis systems (µTAS)”, for biochemical analyses has seen explosive growth over the past decade [6-8]. LOC devices are now widely used in biology and biotechnology [9], DNA and proteins analysis [10, 11], high throughput screening [12], and chemical reactions [13]. Various bio-substances are often analyzed by detecting the immune reaction between antigens in a sample solution or antibodies immobilized on microchannel surfaces using fluorescence detection and the surface plasmon resonance method. In such applications, detection depends significantly on the contact surface. The design and creation of surfaces that have high surface per volume is currently one of the important issues in LOC and µTAS.
Aim to develop a new simple microreactor for bioanalysis, the high density vertically-aligned MWCNTs were synthesized by home-made equipment aided with the chemical vapor deposition (CVD) and studied by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). Also, the wettability of the MWCNTs was investigated after Au coating and plasma irradiation by electron cyclotron resonance (ECR) for potential bioanalysis applications.
Concept of Fabricated 3D Surface by Vertically-aligned MWCNTs for Microreactor Based μTAS
Since CNTs have high-aspect ratios, in micro total analysis systems (μTAS), they can form a much wider reaction field on the surface, which is useful for biological applications [14]. The microreactors in these systems have been proposed to be microchannels or microwells based on the droplets. All of these systems have surfaces with a very high surface-to-volume ratio. From this point-of-view, a reactor with MWCNTs can improve existing microreactor capabilities obviously. In order to achieve a high quality μTAS reactor, synthesis of MWCNTs with a high density is required.
Figure 1 shows the concept of a microreactor with MWCNTs. The relevant proposed method is to locate primary antibodies on the CNTs surface. Thus, primary antibodies are located in three dimensions. Furthermore, the representative size (L in the Figure. 1) will be changed (cf. Figure 1(a) and 1(b)). In a traditional microreactor, L is the depth or width of a microchannel or a microwell. However, L in the new reactor becomes the length between vertically aligned CNTs if the reaction field height is coincident with the CNT length. Consequently, the basic diffusion can be expressed as 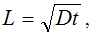 , where D is the diffusion coefficient and t is the diffusion time. By decreasing the value of L, t decreases as well. When the primary antibodies are located in three dimensions, appropriate decreases in the size will lead to higher sensitivities and reaction speed.
, where D is the diffusion coefficient and t is the diffusion time. By decreasing the value of L, t decreases as well. When the primary antibodies are located in three dimensions, appropriate decreases in the size will lead to higher sensitivities and reaction speed.

Figure 1. Concept of a microreactor based μTAS with CNTs.
It is well known that etching-based processes are used for the fabrication of μm~nm size structures. The advantage of etching is that it can be used to fabricate structures with high accuracy. However, it is difficult for etching to make under several nanometers just like CNTs. UV and X-ray lithography can make structures or patterns within 100 nm, but these methods are expensive. On the other hand, synthesis of CNTs currently can be achieved easily at low cost. In this research, MWCNTs are synthesized only by the catalyst pre-deposited by CVD and ethanol with the home-made equipment. Furthermore, CNTs unique structures such as high aspect ratios and densities cannot be fabricated by etching techniques. Therefore, the new microreactor with CNTs proposed in this work will have a promising potential application in μTAS for bioanalysis.
Experimental Procedure
Catalyst coated for synthesizing vertically-aligned MWCNTs
The catalyst (Fe) was deposited on the substrate (mc-Si) by electron cyclotron resonance (ECR), and the related deposition parameters are listed in Table 1.
Table 1. ECR processing parameters for Fe deposition
Substrate |
Si (100) |
Catalyst |
Fe/Al |
Irradiation time |
60, 150, 300, 600 s |
Accelerated voltage |
2,500 V |
Ion current density |
12.0 mA/cm2 |
Gas |
Ar |
Gas flow rate |
0.6 SCCM |
Vacuum |
1.5×10-4 Pa |
MWCNTs synthesis
For vertically-aligned MWCNTs synthesis, the schematic diagram of the home-made equipment is shown in Figure 2. The size of vacuum chamber and the heater is Φ100×150 mm and 50×30 mm respectively. The electrical resistance is taken as the heating resource. The maximum temperature can be reached to 1680 ºC. Ethanol in a ceramic container is located under the heater, and the specimen deposited with the catalyst is placed on the heater in the vacuum chamber. A DC power supplier is applied to heat the specimens in the vacuum (1×10-2 Pa) under 38~40 A.
Figure 2. Schematic diagram of home-made equipment for MWCNTs synthesis
Raman spectroscopy analysis
The characteristics of the vertically aligned MWCNTs synthesized were evaluated by Raman spectroscopy analysis. Table 2 shows the conditions of the Raman spectroscopy analysis. Generally, typical Raman shifts (cm-1) of CNTs include the G-band, the D-band and RBM, where the peak of the G-band is at about 1590 cm-1.and the D-band is 1350 cm-1. The CNTs is evaluated on the basis of the ratio between the G-band and the D-band.
Table 2. Conditions for Raman spectroscopy analysis
Laser wavelength |
532 nm |
Laser power |
30 mW × 0.5% |
Irradiation spot |
5 μm |
Integration times |
30 |
Table 3. Parameters of dice cutting and microreactor
Size of microreactor unit |
100 µm × 100 μm |
Gap among reactors |
35 µm |
Cutting speed |
0.5 mm/s |
Cutting depth |
50 μm |
Microreactor fabrication with CNTs
Results and Discussion
Morphology of synthesized vertically-aligned MWCNTs
The synthesized vertically-aligned MWCNTs are shown in Figure 4. It shows that the general growth direction was normal to the surface. They appeared wiggly due to CNTs tightly packed during growth. The TEM image of one typical CNT is shown in Figure 5. The image reveals that they were definitely multi-walled nanotubes. The diameter was about 3~5 nm. Also, the lengths of the CNTs were estimated to be 10~35 µm. Moreover, the morphology of the relevant microreactor is shown in Figure 6.
Figure 3. Miroreactor fabrication with MWCNTs.
Figure 4. SEM image of synthesized vertically-aligned MWCNTs
Figure 5. TEM image of a CNT.
Figure 6. Microreactor with vertically-aligned MWCNTs
Result of Raman spectroscopy analysis
The result of Raman spectroscopy analysis of the synthesized vertically-aligned MWCNTs is shown in Figure 7. It elucidates that the peak of D-band was higher than that of G-band and the ratio of IG/ID was about 0.75, which indicates that many CNTs were disordered. As the media in the microreactor, the disordered MWCNTs will be inclined to catch the antibody more easily. Therefore, the microreactor with such MWCNTs must be more effective for the practical applications than that of the traditional reactors.
Figure 7. Raman spectra of synthesized MWCNTs
Wettability of the vertically-aligned MWCNTs
The wettability of surface of microreactor with MWCNTs was investigated by the droplet of pure water, and the volume of the droplets was 1 μl. Result shows that the contact angle on the vertically-aligned CNTs film was 146.6º resulted in super-hydrophobic as shown in Figure 8(a), and the related schematic diagram is shown in Figure 8(b). For changing the wettability, the Au coating and plasma irradiation were carried out with the plasma irradiation time of 5 min and ion current of 6.5 mA.
Figure 8. Wettability of the vertically-aligned MWCNTs.
Figure 9 shows the contact angles of the vertically-aligned MWCNTs surface coated with Au and irradiated with the plasma, respectively. Results show that the processed surface changed to a super-hydrophilic surface after Au coating and plasma irradiation. More attractively, the contact angle on the CNTs film irradiated by plasma was close to 0º as shown in Figure 9(c), and its corresponding schematic diagram is shown in Figure 9(d). However, the contact angle of the vertically-aligned MWCNTs surface with Au coating (Figures. 9(a) and (b)) was higher than that of surface with plasma irradiation which indicates the droplet cannot be infiltrated into CNTs completely with Au coating on the surface resulted in the successive application failure.
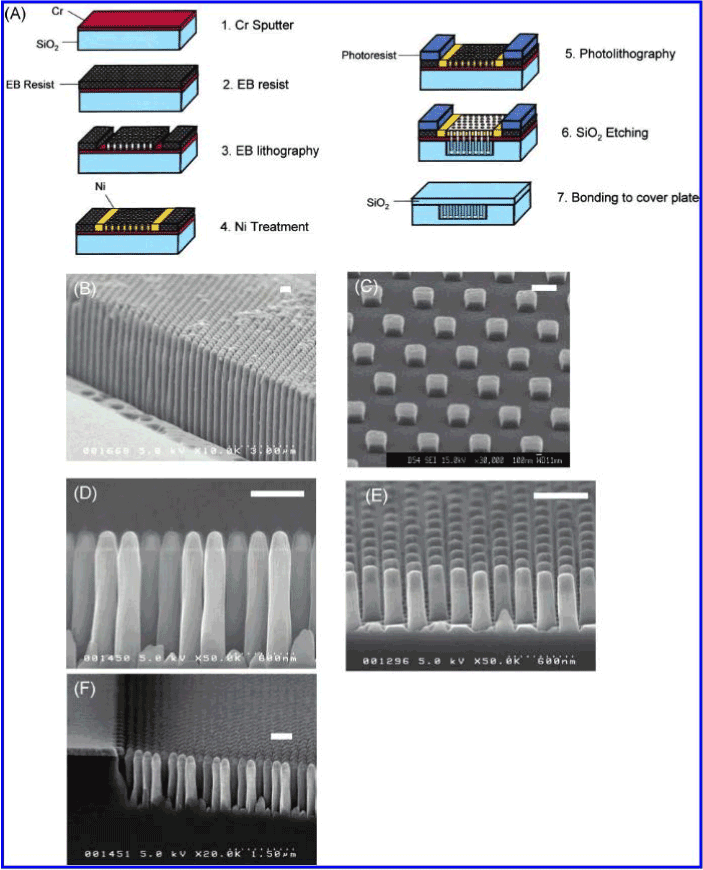
Figure 9. Wettability of the vertically-aligned MWCNTs film with Au coating and plasma irradiation.
Conclusion
The simple microreactor potential for bioanalysis was successfully fabricated with MWCNTs. Results show that the initial synthesized surface with the MWCNTs film behaves as a super-hydrophobic surface. After coating with Au or irradiating by plasma it changes to a super-hydrophilic surface. Moreover, the effect of plasma treatment on the wettability changed from super-hydrophobic to super-hydrophilic is definitely superior to that of Au coating. It indicates that microreactor with surface treated MWCNTs can be more in favor of the potential applications for bioanalysis.
Acknowledgements
The work is supported by the Strategic Research Grant (SRG) from City University of Hong Kong (Grant No.: 7004598).
References
- Shah KA, Tali BA (2016) Synthesis of carbon nanotubes by catalytic chemical vapour deposition: A review on carbon sources, catalysts and substrates. Materials Science in Semiconductor Processing 41: 67-82.
- Zamani P, Higgins DC, Hassan FM, Fu XG, Choi JY, et al. (2016) Highly active and porous graphene encapsulating carbon nanotubes as a non-precious oxygen reduction electrocatalyst for hydrogen-air fuel cells. Nano Energy 26: 267-275.
2021 Copyright OAT. All rights reserv
- Moreno NG, Gervasio D, García AG, Robles JFP (2015) olybenzimidazole-multiwall carbon nanotubes composite membranes for polymer electrolyte membrane fuel cells. Journal of Power Sources 300: 229-237.
- Lawal AT (2016) Synthesis and utilization of carbon nanotubes for fabrication of electrochemical biosensors. Materials Research Bulletin 73: 308-350.
- Rabti A, Raouafi N, Merkoçi A. Bio(Sensing) devices based on ferrocene–functionalized graphene and carbon nanotubes. Carbon 2016; 108: 481-514.
- Lafleur JP, Jönsson A, Senkbeil S, Kutter JP (2016) Recent advances in lab-on-a-chip for biosensing applications. Biosens Bioelectron 76: 213-233. [crossref]
- Temiz Y, Lovchik RD, Kaigala GV, Delamarche E (2015) Lab-on-a-chip devices: How to close and plug the lab? Microelectronic Engineering 132: 156-175.
- Ertl P, Sticker D, Charwat V, Kasper C, Lepperdinger G (2014) Lab-on-a-chip technologies for stem cell analysis. Trends Biotechnol 32: 245-253. [crossref]
- Jastrzebska E, Tomecka E, Jesion I (2016) Heart-on-a-chip based on stem cell biology. Biosens Bioelectron 75: 67-81. [crossref]
- House DL, Chon CH, Creech CB, Skaar EP, Li DQ (2010) Miniature on-chip detection of unpurified methicillin-resistant Staphylococcus aureus (MRSA) DNA using real-time PCR. Journal of Biotechnology 146: 93-99.
- García-Otero N, Barciela-Alonso MC, Moreda-Piñeiro A, Bermejo-Barrera P (2013) Study of extraction procedures for protein analysis in plankton samples by OFFGEL electrophoresis hyphenated with Lab-on-a-chip technology. Talanta 115: 631-641.
- Skardal A, Shupe T, Atala A (2016) Organoid-on-a-chip and body-on-a-chip systems for drug screening and disease modeling. Drug Discov Today 21: 1399-1411. [crossref]
- Mirasoli M, Guardigli M, Michelini E, Roda A (2014) Recent advancements in chemical luminescence-based lab-on-chip and microfluidic platforms for bioanalysis. Journal of Pharmaceutical and Biomedical Analysis 87: 36-52.
- Nakanishi W, Minami K, Shrestha LK, Ji QM, Hill JP, et al. (2014) Bioactive nanocarbon assemblies: Nanoarchitectonics and applications. Nano Today 9: 378-394.









