Abstract
Objectives: The objective of this study was to evaluate the performance of the i-CHROMA™ Point-Of-Care Test (POCT) method for the quantification of Human Chorionic Gonadotrophin (hCG), Leutinzing Hormone (LH) and Follicle Stimulating Hormone (FSH).
Design and Methods: Samples 1-12 of cycles 40 and 41 from Randox International Quality Assessment Scheme (RIQAS) were analysed with the i-CHROMA™ hCG, LH and FSH methods. The estimates were compared with the mean estimates from the other laboratory methods participating in the RIQAS. Results: The i-CHROMA™ hCG, LH and FSH methods compared very well with all the laboratory methods: Abbott Architect, BioMerieux Vidas, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Beckman DXI 600/800, Roche Elecsys, Diasorin Liaison, Monobind Inc ELISA/CLIA, Roche COBAS® 4000/e411, Roche Modular E170, Beckman Access/LXI725, Snibe Maglumi Analysers, and Ortho Vitros 3600/5600/ECi. The laboratory methods with the best combination of a very good correlation (r2 >0.9) and bias (+/- 10% difference) for hCG were: Ortho Vitros 3600/5600/ECi, Snibe Maglumi Analysers, Siemens/DPC Immulite 1000, Siemens Dimension and the bioMerieux, VIDAS / mini VIDAS; for LH were: Diasorin Liaison and Ortho Vitros 3600/5600/ECi; and for FSH were: BioMerieux VIDAS, Siemens Centaur XP/XPT/Classic and Diasorin Liaison.
Conclusion: the i-CHROMA™ hCG, LH and FSH methods compared very well with the laboratory hCG LH and FSH methods enrolled in the RIQAS.
Key words
i-CHROMA, POCT immunoassay, human chorionic gonadotrophin, leutinizing hormone, follicle stimulating hormone
Introduction
Human Chronic Gonadotrophin (hCG), Leutinizing Hormone (LH) and Follicle Stimulating Hormone (FSH) are heterodimeric glycoproteins that are involved in the female reproductive system. Both LH and FSH stimulate follicular growth and maturation, ovulation, and the development of the corpus luteum but hCG on the other hand is secreted from the placenta, and these glycoproteins are used as an indication of ovulation and pregnancy in women [1,2]. In pregnancy, hCG is nearly always detectable in serum and urine within 16 days after ovulation.
Historically, hCG was detected using bioassays, which, after several decades, were replaced with radioimmunoassays [3]. More recently, mass spectrometry–based assays have been developed for the detection and quantification of hCG in serum and urine [4-6]. Now qualitative and quantitative serum human chorionic gonadotropin (hCG) tests are used to diagnose pregnancy. Qualitative tests assess whether or not hCG is present, and quantitative hCG tests detect the amount of hCG, and the isoforms of hCG present, therefore identifying how far along the patient is [7]. One study researched the advantages of both types of hCG testing and concluded that there was no clear advantage in using quantitative hCG tests [8]. However, other studies have found quantitative hCG methods to be much more beneficial than qualitative methods [9].
Currently, there are several qualitative point- of- care rapid test kits for hCG, LH and FSH on the market, however, there are very few quantitative point of care hCG testing devices and very little information on quantitative point of care LH and FSH testing devices. One quantitative point of care hCG testing device is the new Abbott i-STAT hCG device, which when compared with the Abbott Architect Ci8200, showed a very good correlation (r2 = 0.994) [10]. Another study comparing the i-STAT hCG methods with three other laboratory methods (Abbott Architect Total β-hCG; Beckman Dxl Total β-hCG ; and Roche Cobas e601 hCG+β) also showed that the i-STAT results agreed most closely with the Abbott Architect Total β-hCG assay and greater differences were observed with Beckman Dxl Total β-hCG and Roche Cobas e601 hCG+β assays (mean differences 9.3% and 12.3%, respectively) [11]. We decided to compare the performance of a novel quantitative point of care test device, the i-CHROMA™ hCG, LH and FSH methods against a variety of other laboratory hCG, LH and FSH methods participating in the Randox International Quality Assessment Scheme (RIQAS).
Materials and methods
hCG materials –RIQAS
Samples 1-12 of cycles 40 and 41 from the RIQAS scheme were reconstituted and analysed for total hCG using the i-CHROMA™ hCG method described using the i-CHROMA™ hCG concentration estimations. There were 12 laboratory methods registered with this scheme that had provided estimates of hCG for samples 1-12 of cycles 40 and 41 that had an n value of greater than 23: Abbott Architect (n=24), BioMerieux Vidas (n=24), Roche hCG + Beta (n=24), Siemens Centaur XP/XPT/Classic (n=24), Siemens Dimension (n=24), Siemens/DPC Immulite 2000 (n=24), Siemens/DPC Immulite 1000 (n=24), Beckman DXI 600/800 (n=24), Roche hCG Stat (n=24), Beckman Access/LXI725 (n=24), Snibe Maglumi Analysers (n=23), and Ortho Vitros 3600/5600/ECi (n=24).
LH materials – RIQAS
Samples 1-12 of cycles 40 and 41 from the RIQAS scheme were reconstituted and analysed for LH using the i-CHROMA™ LH method described using the i-CHROMA™ LH concentration estimations. There were 15 laboratory methods registered with this scheme that had provided estimates of LH for samples 1-12 of cycles 40 and 41 that had an n value of greater than 22: Abbott Architect (n=24), BioMerieux Vidas (n=24), Roche COBAS® 6000/8000 (n=24), Siemens Centaur XP/XPT/Classic (n=24), Siemens/DPC Immulite 2000 (n=24), Siemens/DPC Immulite 1000 (n=24), Beckman DXI 600/800 (n=24), Roche Elecsys (n=24), Diasorin Liaison (n=23), Monobind Inc ELISA/CLIA (n=22), Roche COBAS® 4000/e411 (n=24), Roche Modular E170 (n=24), Beckman Access/LXI725 (n=24), Snibe Maglumi Analysers (n=24), and Ortho Vitros 3600/5600/ECi (n=24).
FSH materials – RIQAS
Samples 1-12 of cycles 40 and 41 from the RIQAS scheme were reconstituted and analysed for FSH using the i-CHROMA™ FSH method described using the i-CHROMA™ FSH concentration estimations. There were 15 laboratory methods registered with this scheme that had provided estimates of FSH for samples 1-12 of cycles 40 and 41 that had an n value of greater than 23: Abbott architect (n=24), BioMerieux Vidas (n=24), Roche COBAS® 6000/8000 (n=24), Siemens Centaur XP/XPT/Classic (n=24), Siemens/DPC Immulite 2000 (n=24), Siemens/DPC Immulite 1000 (n=24), Beckman DXI 600/800 (n=24), Roche Elecsys (n=24), Diasorin Liaison (n=24), Monobind Inc ELISA/CLIA (n=23), Roche COBAS® 4000/e411 (n=24), Roche Modular E170 (n=24), Beckman Access/LXI725 (n=24), Snibe Maglumi Analysers (n=24), and Ortho Vitros 3600/5600/ECi (n=24) .
Methods
i-CHROMA™ assay principle
The i-CHROMA™ system is a quantitative immunoassay system based on a fluorescence immunoassay technology. The i-CHROMA™ assay systems uses a sandwich immunodetection method, such that the fluorescence labelled detector antibody or antigen binds to the target protein in sample (sample mixed with a pre-measured amount of detection buffer containing fluorescence labelled anti-target protein monoclonal antibodies and anti-rabbit IgG). The mixture is then loaded onto the well of the test strip, the target protein complexes are immobilised on the matrix by anti-target protein bound to the matrix and after an incubation time period of immune reaction the test and control lines are scanned for fluorescence intensity. The fluorescence intensities converted into a P - target protein concentration calculated by pre-programmed calibration process. The result of the test is displayed on the reader as milli-International Units per millilitre (mU/ml). The i-CHROMA™ total hCG, LH and FSH methods are all CE certified.
i-CHROMA™ hCG concentration estimations
Seventy five microliters (75 µL) of reconstituted RIQAS sample was collected, and added into the tube containing detection buffer containing fluorescence labelled anti-HCG monoclonal antibodies and anti-rabbit IgG. The tube was shaken up and down 10 times or more and 75 µl of the mixture collected and transferred onto the sample well of the test device cartridge, the test device cartridge was left to incubate at room temperature for 15 minutes. Then the test device cartridge was placed into the test device holder of the i-CHROMA™ reader, the “select” button pressed and the results read off the display screen.
i-CHROMA™ LH concentration estimations
Seventy-five microliters (75 µL) of reconstituted RIQAS sample was collected, and added into the tube containing detection buffer containing fluorescence labelled anti-LH monoclonal antibodies and anti-rabbit IgG. The tube was shaken up and down 10 times or more and 75 µl of the mixture collected and transferred onto the sample well of the test device cartridge, the test device cartridge was left to incubate at room temperature for 15 minutes. Then the test device cartridge was placed into the test device holder of the i-CHROMA™ reader, the “select” button pressed and the results read off the display screen.
i-CHROMA™ FSH concentration estimations
One hundred and fifty microliters (150 µL) of reconstituted RIQAS sample was collected, and added into the tube containing detection buffer containing fluorescence labelled anti-FSH monoclonal antibodies and anti-rabbit IgG. The tube is shaken up and down 10 times or more and 75 µl of the mixture collected and transferred onto the sample well of the test device cartridge, the test device cartridge is left to incubate at room temperature for 15 minutes. Then the test device cartridge was placed into the test device holder of the i-CHROMA™ reader, the “select” button pressed and the results read off the display screen.
Statistics and ethics approval
The results of the 24 samples from cycles 40 and 41 of hCG, LH and FSH RIQAS using the i-CHROMA™ methods were compared with the mean results of the laboratory methods in the RIQAS database using Correlation coefficient, Bland-Altman plots and Paired T-tests. There samples used in this study were external quality control samples purchased from RIQAS. Therefore, no informed consent was necessary.
Results
hCG RIQAS – Correlation and bias
The results of the total hCG i-CHROMA™ method correlated well with all methods: Abbott Architect (r2 = 0.9976), BioMerieux Vidas/mini VIDAS (r2 = 0.9955), Roche hCG + Beta (r2 = 0.9983), Siemens Centaur XP/XPT/Classic (r2 = 0.9938), Siemens Dimension (r2 = 0.9971), Siemens/DPC Immulite 2000 (r2 = 0.9976), Siemens/DPC Immulite 1000 (r2 = 0.9969), Beckman DXI 600/800 (r2 = 0.9977), Roche hCG Stat (intact) (r2 = 0.9961), Beckman Access/LXI725 (r2 = 0.9978), Maglumi Analysers (r2 = 0.9976) and Ortho Vitros 3600/5600/ECi (r2 = 0.9985) (Table 1). The bias ranged between -26.5 mU/ml (14.88% bias difference) and +97.6 mU/ml (54.74% bias difference). The laboratory methods with the best combination of a very good correlation (r2> 0.9) and bias (+/- 10% difference) with the i-CHROMA™ hCG method were the Ortho Vitros 3600/5600/ECi (Figure 1), bioMerieux, Vidas/mini VIDAS, Siemens Dimension, Siemens/DPC Immulite 1000 and Snibe Maglumi analysers. The results of the paired t-test show that nine out of twelve methods had a p value of greater than 0.05: bioMerieux, VIDAS / mini VIDAS, Roche hCG+Beta, Siemens Centaur XP/XPT/Classic, Siemens Dimension, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Roche hCG STAT (Intact), Snibe Maglumi analysers and Ortho Vitros 3600/5600/ECi. Three of the twelve methods had a p value of less than 0.05: Abbott Architect, Beckman DxI 600/800 and Beckman Access/LXi725. These results signify a significant difference between the aforementioned three methods and the i-CHROMA™ hCG method. The box and whisker plots show the distribution of the results of all the hCG methods (Figure 2).
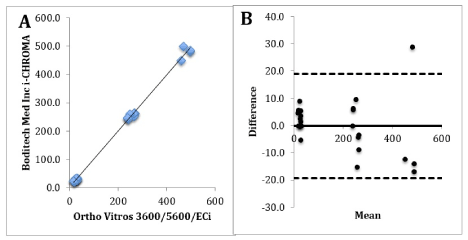
Figure 1A. Correlation graph of i-CHROMA™ vs Ortho Vitros 3600/5600/ECi. 1B: Bland-Altman difference plot of i-CHROMA™ vs Ortho Vitros 3600/5600/ECi.
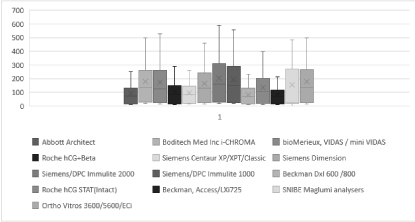
Figure 2. Box and Whisker plots showing the distribution of results (concentration mU/ml) for all the hCG assays tested.
Table 1. Correlation coefficient, bias and p values for distribution 40 and 41 hCG RIQAS samples.
Method |
R value |
Bias (mU/ml) |
Bias Difference (%) |
P Value |
n |
Abbott Architect |
0.9976 |
86.2 |
48.37 |
0.03 |
24 |
bioMerieux, VIDAS / mini VIDAS |
0.9955 |
3.1 |
1.72 |
0.95 |
24 |
Roche hCG+Beta |
0.9983 |
72.7 |
40.78 |
0.08 |
24 |
Siemens Centaur XP/XPT/Classic |
0.9938 |
79.2 |
44.42 |
0.05 |
24 |
Siemens Dimension |
0.9971 |
13.6 |
7.61 |
0.78 |
24 |
Siemens/DPC Immulite 2000 |
0.9976 |
-26.5 |
-14.88 |
0.62 |
24 |
Siemens/DPC Immulite 1000 |
0.9969 |
-17.6 |
-9.85 |
0.74 |
24 |
Beckman DxI 600 /800 |
0.9977 |
93.5 |
52.44 |
0.02 |
24 |
Roche hCG STAT (Intact) |
0.9961 |
40.4 |
22.67 |
0.37 |
24 |
Beckman, Access/LXi725 |
0.9978 |
97.6 |
54.74 |
0.01 |
24 |
Snibe Maglumi analysers |
0.9976 |
2.8 |
1.67 |
0.96 |
23 |
Ortho Vitros 3600/5600/ECi |
0.9985 |
-0.2 |
-0.11 |
0.99 |
24 |
LH RIQAS – Correlation and bias
The results of the LH i-CHROMA™s method correlated well with all methods: Abbott Architect (r2 = 0.933), BioMerieux Vidas/mini VIDAS (r2 = 0.924), Roche COBAS® 6000/8000 (r2 = 0.944), Siemens Centaur XP/XPT/Classic (r2 = 0.914), Siemens/DPC Immulite 2000 (r2 = 0.980), Siemens/DPC Immulite 1000 (r2 = 0.959), Beckman DXI 600/800 (r2 = 0.937), Roche Elecsys (r2 = 0.944), Diasorin Liaison (r2 = 0.956), Monobind Inc ELISA/CLIA (r2 = 0.958), Roche COBAS® 4000/e411 (r2 = 0.943), Roche Modular E170 (r2 = 0.941), Beckman Access/LXI725 (r2 = 0.925), Snibe Maglumi Analysers (r2 = 0.974) and Ortho Vitros 3600/5600/ECi (r2 = 0.983) (Table 2). The bias ranged between -10.4 mU/ml (-26.6% bias difference) and +19.1 mU/ml (48.7% bias difference).
Table 2. Correlation coefficient, bias and p values for distribution 40 and 41 LH RIQAS samples.
Method |
R value |
Bias (mU/ml) |
Bias Difference (%) |
P Value |
n |
Abbott Architect |
0.933 |
16.2 |
41.4 |
0.004 |
24 |
bioMerieux, VIDAS / mini VIDAS |
0.924 |
14.2 |
36.3 |
0.014 |
24 |
Roche COBAS® 6000/8000 |
0.944 |
12.1 |
31.0 |
0.033 |
24 |
Siemens Centaur XP/XPT/Classic |
0.914 |
12.7 |
32.3 |
0.028 |
24 |
Siemens/DPC Immulite 2000 |
0.980 |
-8.6 |
-21.9 |
0.228 |
24 |
Siemens/DPC Immulite 1000 |
0.959 |
-10.4 |
-26.6 |
0.162 |
24 |
Beckman DxI 600 /800 |
0.937 |
19.1 |
48.7 |
0.0006 |
24 |
Roche Elecsys |
0.944 |
11.9 |
30.5 |
0.037 |
24 |
Diasorin Liaison |
0.956 |
3.6 |
8.9 |
0.558 |
23 |
Monobind Inc ELISA/CLIA |
0.958 |
-5.1 |
-12.9 |
0.461 |
23 |
Roche COBAS® 4000/e411 |
0.943 |
12.2 |
31.1 |
0.033 |
24 |
Roche Modular E170 |
0.941 |
12.3 |
31.5 |
0.030 |
24 |
Beckman, Access/LXi725 |
0.925 |
18.2 |
46.6 |
0.001 |
24 |
Snibe Maglumi analysers |
0.974 |
4.9 |
12.6 |
0.424 |
24 |
Ortho Vitros 3600/5600/ECi |
0.983 |
0.2 |
0.42 |
0.978 |
24 |
The laboratory methods with the best combination of a very good correlation (r2>0.9) and bias (+/- 10% difference) with the i-CHROMA™ LH method were the Ortho Vitros 3600/5600/ECi (Figure 3) and Diasorin Liaison. The results of the paired t-test show that six out of the fifteen methods had a p value of greater than 0.05: Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Diasorin Liaison, Monobind Inc ELISA/CLIA, Snibe Maglumi analysers and Ortho Vitros 3600/5600/ECi. BioMerieux Vidas/mini VIDAS, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Beckman DXI 600/800, Roche Elecsys, Roche COBAS® 4000/e411, Roche Modular E170 and Beckman Access/LXi725. These results signify a significant difference between the afore mentioned nine methods and the i-CHROMA™ LH method. The box and whisker plots show the distribution of the results of all the LH methods (Figure 4-5).
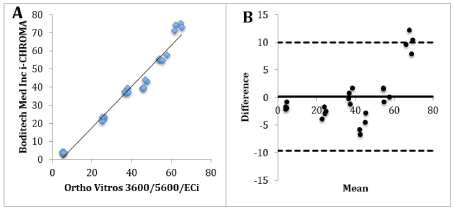
Figure 3A. Correlation graph of i-CHROMA™ vs Ortho Vitros 3600/5600/ECi. 3B: Bland-Altman difference plot of i-CHROMA™ vs Ortho Vitros 3600/5600/ECi.
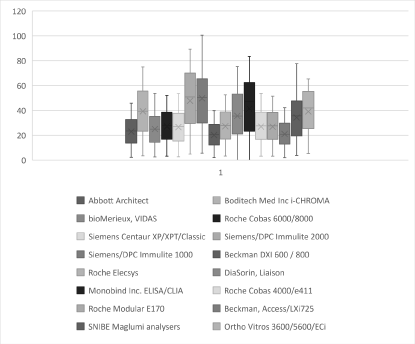
Figure 4. Box and Whisker plots showing the distribution of results (concentration mU/ml) for all the LH assays tested.
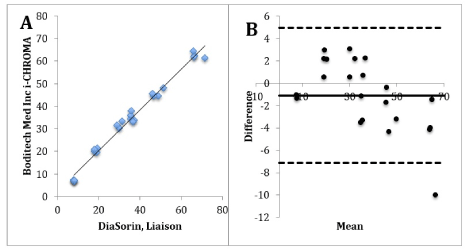
Figure 5A. Correlation graph of i-CHROMA™ vs Diasorin Liaison. 5B: Bland-Altman difference plot of i-CHROMA™ vs Diasorin Liaison.
FSH RIQAS – Correlation and bias
The results of the FSH i-CHROMA™s method correlated well with all methods: Abbott Architect (r2 = 0.997), BioMerieux Vidas/mini VIDAS (r2 = 0.995), Roche COBAS® 6000/8000 (r2 = 0.997), Siemens Centaur XP/XPT/Classic (r2 = 0.991), Siemens/DPC Immulite 2000 (r2 = 0.992), Siemens/DPC Immulite 1000 (r2 = 0.993), Beckman DXI 600/800 (r2 = 0.997), Roche Elecsys (r2 = 0.997), Diasorin Liaison (r2 = 0.992), Monobind Inc ELISA/CLIA (r2 = 0.960), Roche COBAS® 4000/e411 (r2 = 0.997), Roche Modular E170 (r2 = 0.997), Beckman Access/LXI725 (r2 = 0.991), Snibe Maglumi Analysers (r2 = 0.940) and Ortho Vitros 3600/5600/ECi (r2 = 0.996) (Table 3).
Table 3. Correlation coefficient, bias and p values for distribution 40 and 41 FSH RIQAS samples.
Method |
R value |
Bias (mU/ml) |
Bias Difference (%) |
P Value |
n |
Abbott Architect |
0.997 |
9.8 |
29.0 |
0.034 |
24 |
bioMerieux, VIDAS / mini VIDAS |
0.995 |
1.5 |
4.5 |
0.772 |
24 |
Roche COBAS® 6000/8000 |
0.997 |
7.6 |
22.4 |
0.111 |
24 |
Siemens Centaur XP/XPT/Classic |
0.991 |
1.8 |
5.4 |
0.729 |
24 |
Siemens/DPC Immulite 2000 |
0.992 |
6.5 |
19.0 |
0.177 |
24 |
Siemens/DPC Immulite 1000 |
0.993 |
7.0 |
20.1 |
0.141 |
24 |
Beckman DxI 600 /800 |
0.997 |
5.6 |
16.5 |
0.248 |
24 |
Roche Elecsys |
0.997 |
6.0 |
17.6 |
0.218 |
24 |
Diasorin Liaison |
0.992 |
-1.1 |
-3.1 |
0.846 |
24 |
Monobind Inc ELISA/CLIA |
0.960 |
9.0 |
25.8 |
0.047 |
23 |
Roche COBAS® 4000/e411 |
0.997 |
5.6 |
16.4 |
0.253 |
24 |
Roche Modular E170 |
0.997 |
7.9 |
23.1 |
0.098 |
24 |
Beckman, Access/LXi725 |
0.991 |
4.8 |
14.0 |
0.333 |
24 |
Snibe Maglumi analysers |
0.940 |
8.9 |
15.2 |
0.042 |
24 |
Ortho Vitros 3600/5600/ECi |
0.996 |
12.1 |
21.7 |
0.008 |
24 |
2021 Copyright OAT. All rights reserv
The bias ranged between -1.1 mU/ml (-3.1% bias difference) and +12.1 mU/ml (21.7% bias difference). The results of the paired t-test show that eleven out of the fifteen methods had a p value of greater than 0.05: BioMerieux Vidas/mini VIDAS, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Beckman DXI 600/800, Roche Elecsys, Diasorin Liaison, Roche COBAS® 4000/e411, Roche Modular E170 and Beckman Access/LXI725. Four of the fifteen methods had a p value of less than 0.05: Abbott Architect, Monobind Inc ELISA/CLIA, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi. These results signify a significant difference between the aforementioned four methods and the i-CHROMA™ FSH method. The box and whisker plots show the distribution of the results of all the FSH methods (Figure 6).
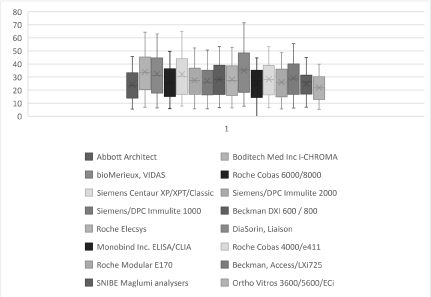
Figure 6. Box and Whisker plots showing the distribution of results (concentration mU/ml) for all the FSH assays tested.
The laboratory methods with the best combination of a very good correlation (r2 > 0.9) and bias (+/- 10% difference) with the i-CHROMA™ FSH method were the Diasorin Liaison (Figure 3), BioMerieux Vidas/Mini VIDAS and Siemens Centaur XP/XPT/Classic.
Discussion
The results of the i-CHROMA™s total hCG method correlated well with all methods: Abbott Architect, BioMerieux Vidas, Roche hCG + Beta, Siemens Centaur XP/XPT/Classic, Siemens Dimension, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Beckman DXI 600/800, Roche hCG Stat (intact), Beckman Access/LXI725, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi. The bias ranged between -26.5 mU/ml (14.88% bias difference) and +97.6 mU/ml (54.74% bias difference). The laboratory methods with the best combination of a very good correlation (r2> 0.9) and bias (+/- 10% difference) with the i-CHROMA™ hCG method were the Ortho Vitros 3600/5600/ECi, bioMerieux, VIDAS / mini VIDAS, Siemens Dimension, Siemens/DPC Immulite 1000 and Snibe Maglumi analysers.
The i-CHROMA™ hCG method reported higher hCG values when compared with nine out of twelve (75%) of the methods: Abbott Architect, BioMerieux Vidas/mini VIDAS, Roche hCG + Beta, Siemens Centaur XP/XPT/Classic, Siemens Dimension, Beckman DXI 600/800, Roche hCG Stat (intact), Beckman Access/LXI725 and Snibe Maglumi Analysers. The i-CHROMA™ hCG method reported lower hCG values when compared to three out of the twelve (25%) methods: Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000 and the Ortho Vitros 3600/5600/ECi. These results are similar to the findings with another quantitative POCT hCG device where the study carried out by Sowder et al. [11] compared the i-STAT β hCG method with the Abbott Architect Total β-hCG, Beckman Dxl Total β-hCG, and Roche Cobas e601 hCG+β, and showed that the i-STAT β hCG method also produced higher results than these methods: Beckman Dxl Total βhCG and Roche Cobas e601 hCG + β assays (mean differences 9.3% and 12.3%, respectively). In our study, the i-CHROMA™ hCG method reported higher values compared to these methods: Beckman DXI 600/800 and Roche hCG+ Beta but with higher mean differences, 52.4% and 40.8%, respectively. The i-CHROMA™ hCG method produced lower values when compared with three of the laboratory hCG methods: Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000 and Ortho Vitros 3600/5600/ECi. This observation is consistent with the findings, which indicate that the Siemens Immulite Series (1000 and 2000) report higher values for total hCG as they detect both intact hCG and free βhCG [12]. Furthermore, the Siemens Immulite method has been described to detect all the hCG structural variants [13] and therefore is not surprising that this method had the greatest difference of -14.88% and -9.85% compared with the i-CHROMA™ hCG method. While the Siemens/DPC Immulite method detects all the hCG variants, the Ortho Vitros ECi method does not detect hCGβcf and poorly detects hCGβ and nicked hCG. The results of the paired t-test show that three of the twelve methods had a p value of less than 0.05: Abbott Architect, Beckman DxI 600/800 and Beckman Access/LXi725, signifying that there might be a significant difference between the results of these methods and the i-CHROMA™ hCG method.
The i-CHROMA™ LH method correlated well with all methods: Abbott Architect (r2 = 0.933), BioMerieux Vidas, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Beckman DXI 600/800, Roche Elecsys, Diasorin Liaison, Monobind Inc ELISA/CLIA, Roche COBAS® 4000/e411, Roche Modular E170, Beckman Access/LXI725, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi. These results are consistent with the comparability studies in the product information sheet, which found that the i-CHROMA™ showed a good correlation with the Beckman Access LH method (0.994) [14]. The bias ranged between -10.4 mU/ml (-26.6% bias difference) and +19.1 mU/ml (48.7% bias difference). The i-CHROMA™ LH method reported higher LH values when compared with twelve out of the fifteen (80%) of the methods: Abbott Architect, BioMerieux Vidas/mini VIDAS, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Beckman DXI 600/800, Roche Elecsys, Diasorin Liaison, Roche COBAS® 4000/e411, Roche Modular E170, Beckman Access/LXI725, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi. The i-CHROMA™ LH method reported lower LH values when compared to three out of the fifteen (20%) methods: Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000 and the Monobind Inc ELISA/CLIA. The laboratory methods with the best combination of a very good correlation (r2>0.9) and bias (+/- 10% difference) with the i-CHROMA™ LH method were the Ortho Vitros 3600/5600/ECi and Diasorin Liaison. Despite the very good correlation nine of the fifteen methods: Abbott Architect, BioMerieux Vidas, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Beckman DXI 600/800, Roche Elecsys, Roche COBAS® 4000/e411, Roche Modular E170 and Beckman Access/LXi725, showed there was a significant difference between the results of these methods and the i-CHROMA™ LH method.
The results of the i-CHROMA™ FSH method correlated well with all methods: Abbott Architect, BioMerieux Vidas, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Beckman DXI 600/800, Roche Elecsys, Diasorin Liaison, Monobind Inc ELISA/CLIA, Roche COBAS® 4000/e411, Roche Modular E170, Beckman Access/LXI725, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi. These results are consistent with the comparability studies in the product information sheet, which found that the i-CHROMA™ showed a very good correlation with the Biomerieux Vidas/mini VIDAS method (r2 = 0.9914) [15]. The bias ranged between -1.1 mU/ml (-3.1% bias difference) and +12.1 mU/ml (21.7% bias difference). The i-CHROMA™ FSH method reported higher hCG values when compared with fourteen out of the fifteen (93%) of the methods: Abbott Architect, BioMerieux Vidas/mini VIDAS, Roche COBAS® 6000/8000, Siemens Centaur XP/XPT/Classic, Siemens/DPC Immulite 2000, Siemens/DPC Immulite 1000, Beckman DXI 600/800, Roche Elecsys, Monobind Inc ELISA/CLIA, Roche COBAS® 4000/e411, Roche Modular E170, Beckman Access/LXI725, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi. The i-CHROMA™ FSH method reported lower hCG values when compared to one out of the fifteen (7%) methods: the Diasorin Liaison. The laboratory methods with the best combination of a very good correlation (r2>0.9) and bias (+/- 10% difference) with the i-CHROMA™ FSH method were the Diasorin Liaison, BioMerieux Vidas/Mini VIDAS and Siemens Centaur XP/XPT/Classic. Despite the very good correlation four of the fifteen methods: Abbott Architect, Monobind Inc ELISA/CLIA, Snibe Maglumi Analysers and Ortho Vitros 3600/5600/ECi showed there was a significant difference between the results of these methods and the i-CHROMA™ FSH method.
In summary, the i-CHROMA™ hCG, LH and FSH methods compared very well with the laboratory methods enrolled in the RIQAS. The methods that compared best in terms of correlation (r2>0.9) and bias within +/-10% bias difference) for hCG and LH was the Ortho Vitros 3600/5600/ECi, and the Diasorin Liaison for FSH. Therefore, the POCT i-CHROMA™ hCG, LH and FSH methods could be considered for the quantitative estimation in the primary care and secondary care setting.
References
- Ezcurra D, Humaidan P (2014) A review of luteinising hormone and human chorionic gonadotropin when used in assisted reproductive technology. Reprod Biol Endocrinol 12: 95. [Crossref]
- Hillier SG (2001) Gonadotropic control of ovarian foliicular growth and development. Mol Cell Endocrinol 179: 39-46. [Crossref]
- Braunstein GD (2014) The long gestation of the modern home pregnancy test. Clin Chem 60: 18-21. [Crossref]
- Woldemariam GA, Butch AW (2014) Immunoextraction-tandem mass spectrometry method for measuring intact human chorionic gonadotropin, free beta-subunit, and beta-subunit core fragment in urine. Clin Chem 60: 1089-1097. [Crossref]
- Lund H, Paus E, Berger P, Stenman UH, Torcellini T, et al. (2014) Epitope analysis and detection of human chorionic gonadotropin (hCG) variants by monoclonal antibodies and mass spectrometry. Tumour Biol 35: 1013-1022. [Crossref]
- Lund H, Snilsberg AH, Halvorsen TG, Hemmersbach P, Reubsaet L (2013) Comparison of newly developed immuno-MS method with existing DELFIA® immunoassay for human chorionic gonadotropin determination in doping analysis. Bioanalysis 5: 623-630. [Crossref]
- Kovalevskaya G, Birken S, Kakuma T, O'Connor JF (1999) Early pregnancy human chorionic gonadotropin (hCG) isoforms measured by an immunometric assay for choriocarcinoma-like hCG. J Endocrinol 161: 99-106. [Crossref]
- Furtado L, Lehman C, Thompson C, Grenache D (2012) Should the qualitative serum pregnancy test be considered obsolete? Am J Clin Pathol 137: 194-202. [Crossref]
- Grossman D, Berdichevsky K, Larrea F, Beltran J (2007) Accuracy of a semi-quantitative urine pregnancy test compared to serum beta-hCG measurement: a possible screening tool for ongoing pregnancy after medication abortion. Contraception 76: 101-104. [Crossref]
- Wikstrom AK, Hagmar M, Ronquist G, Larsson A (2015) Evaluation of a plasma hCG method for point of care testing with the aim of shortening test turnaround times. Open J Obstet Gynaecol 5: 341 - 343.
- Sowder AM, Yarbrough ML, Nerenz RD, Mitsios JV, Mortenson R, et al. (446) Analytical performance evaluation of the i-STAT total beta-human chorionic gonadotrophin immunoassay. Clin Chim Acta 446: 165-170. [Crossref]
- Von Lode P, Rainaho J, Pettersson K (2004) Quantitative, wide range, 5 minute point of care immunoassay for total human chorionic gonadotrophin in whole blood. Clin Chem 50: 1026 -1035.
- Cao Z, Rej R (2008) Are laboratories reporting serum quantitative hCG results correctly. Clin Chem 54: 761-764. [Crossref]
- Boditech Med Inc. I-CHROMA LH. 2014 October 16
- Boditech Med Inc. I-CHROMA FSH. 2014 July 7






