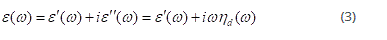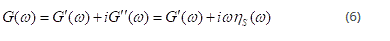Interfacial rheology is an important factor for the stability of emulsions and thin-liquid films containing proteins [1]. Proteins may stabilize interfaces by forming viscoelastic networks that can damp film-stretching and interfacial fluctuations, thus diminishing membrane-thinning [2]. These material properties can be characterized by dilatational and shear surface viscosities, and the respective elastic moduli [3]. We previously used the Langmuir trough and Wilhelmy plate tensiometer in a non-standard way. By means of sinusoidal perturbations (Figure 1) in insulin monolayers we could measure the 2d deformation of the nanofilms [4]. In this commentary we explain the methodology.
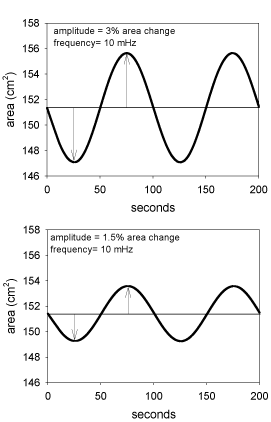
Figure 1. For sinusoidal compression/expansion experiments it is important to check that the area change is really sinusoidal. At least the KSV balances (KSV Instruments, Helsinki, Finland) manufactured 15 years ago did not a real sinusoidal change of the area, but a saw-tooth one. The figure shows a perfect sinusoidal change in the area of the through.
The monolayers must be compressed isometrically up to the desired Π. Then, sinusoidal area perturbations were carried out while simultaneously measuring both Π and phase-shift in response to area changes [3] (Figure 2).
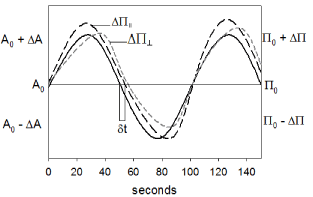
Figure 2. The surface pressure recorded as function of time, as the area (black continuous line) is changed by oscillatory barrier measurements. The surface pressure is given in two orthogonal orientations, (ΔΠ║) and (ΔΠ┴) with respect to the compression direction. Note that ΔΠ║ > ΔΠ┴ indicating a shear modulus.
Since the deformation created by the moving barriers is uniaxial, it contains a superposition of well-defined dilatation and shear. Thus, as the interfacial pressure is a tensor quantity, the values depend on the direction along which the stress is acting [1]. Therefore, if two Wilhelmy plates are placed in orthogonal positions, the following equations describe the total viscoelastic response of the system:

where ε is the (complex) dilatational compressibility, G is the (complex) shear modulus and ΔΠ║ and ΔΠ⏊ are the lateral pressure response registered by the sensing plates placed parallel and perpendicular to the barriers, respectively. If the layer is purely elastic, then area and Π should always be in phase; if it is purely viscous, both parameters should be 90◦ out of phase [3]. Usually, the frequency dependent complex modulus ε(ω) is defined as:
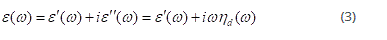
where ε´(ω) and ωηd(ω) are the dilatational elasticity and the dilatational viscosity, respectively. The real part is the elastic (storage) component of the modulus:

and the imaginary part is the dissipative (loss) component:

The phase angle φ is equal to 2πωδt.
The shear elastic modulus G is defined as the ratio between the increase of shear strain and shear stress; the shear viscosity ηs is the ratio between the shear stress and the rate of shear. In an oscillatory measurement one can measure the complex shear modulus as follows:
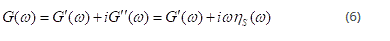
Where the real part is the elastic (storage) component of the modulus:

and the imaginary part is the dissipative (loss) component:

The method developed by Cicuta and Terentjev [3] is useful to characterize the mechanical properties of Langmuir monolayers, by measuring both the compression and shear modulus in a single experiment. The frequency range accessible in KSV instruments is relatively narrow (0.01<ω<0.1 Hz) but is comparable to other surface rheometers. By using this technique, we studied the interfacial behavior of insulins. The response of the nanofilms were critically affected by Zn2+ in the subphase. This cation induced a condensed-like behavior in the compression isotherms, marked variations of the surface electrostatics and considerable hysteresis of the monolayer organization. The technique described in this commentary allowed us to determine the viscoelastic networks of the nanofilms, indicating a behavior resembling a nominal solid [4].
- Petkov JT, Gurkov TD, Campbell BE, Borwankar RP (2000) Dilatational and Shear Elasticity of Gel-like Protein Layers on Air/Water Interface. Langmuir 16: 3703-3711.
- Wilde P, Mackie A, Husband F, Gunning P, Morris V (2004) Proteins and emulsifiers at liquid interfaces. Adv Colloid Interface Sci 108-109: 63-71. [Crossref]
- Cicuta P, Terentjev E (2005) Viscoelasticity of a protein monolayer from anisotropic surface pressure measurements. Eur Phys J E Soft Matter 16: 147-158. [Crossref]
- Grasso E, Oliveira R, Maggio B (2014) Rheological properties of regular insulin and aspart insulin Langmuir monolayers at the air/water interface: condensing effect of Zn2+ in the subphase. Colloids Surf B Biointerfaces 115: 219-228. [Crossref]