Introduction
Fragile chromosome X syndrome is the prime cause of hereditary mental retardation and the second most frequent chromosomopathy after Down’s syndrome [1,2]. It is estimated to affect one in 4000 males and that there is one female carrier per 800 and one male carrier per 5000 [3,4]. The inactivated gene FMR1 codifies the disease and is altered at locus Xq27.3 (long arm of chromosome X) [1,5]. This gene determines the non-production of the protein FMRP [2,6,7] the deficiency of which causes the disease, as this protein is essential in the regulation of neuronal changes, stimuli, and maturation — that is, of the development of the nervous system — and also affects the development of connective tissue [3], the cause of the articular hyperlaxity presented by these patients [2].
The discovery of the gene FMR1 in 1991 led to a growth in DNA-based studies for diagnosis [3]. Both carriers and sufferers are detected. In 1995, it was developed a laboratory technique to identify anti-protein FMRP antibodies in lymphocytes which are useful for the initial screening of males with mental retardation of unknown etiology [6]. The technique of FMRP protein detection is currently used successfully in both prenatal (chorionic villi and amniotic fluid) and postnatal (blood cells and hair bulb) diagnosis [2,6].
Varying degrees of chromosomal alteration in fragile chromosome X syndrome have been described that are seen as physical and psychological effects. The most-frequent physical characteristics of these children are elongated and narrow face, prominent mandible, macro-orchism, prolapsed mitral valve, large and prominent ears, strabismus, otitis of the middle ear, excessive pronation of the foot, and also increased articular laxity [2,3,5]. The most-frequent psychological characteristics are hyperactivity, attention disorders, autistic behavior in some cases, extreme shyness, repetitive language, a tendency to avoid direct eye contact, typical flapping of the hands during expression, nervousness and hypersensitivity to stimuli (which the authors emphasize as being important for the understanding of the case described below), resistance to change of any sort, behavioral problems and aggressiveness in some cases [2-4].
Sequelae in the foot
The authors’ experience with this type of patient is in twelve male cases. The common pattern observed in these children’s feet has been valgus of the hindfoot secondary to articular hyperlaxity, with values of resting calcaneal stance position over 10° valgus, and a longitudinal cleft in the plantar skin between the second and third metatarsals, not reported in the literature (Figure 1). Only twelve cases examined by the authors are not enough to establish the presence of this cleft as a pathognomonic sign. However authors stress this finding because appeared in 100% of the cases. It would be necessary further studies to validate this sign as a diagnostic sign.
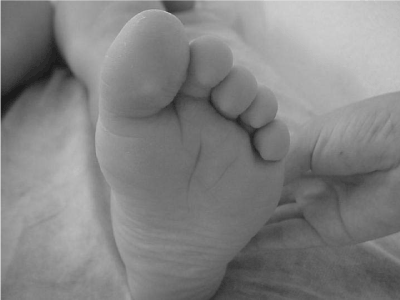
Figure 1. The intermetatarsal plantar cleft shown in the photo was seen in the feet of the twelve patients with fragile chromosome x syndrome who had been attended by the authors along their careers.
Case report
A 18-year-old patient with fragile chromosome X syndrome presented at our podiatric center in Seville (Spain), 1.80 m tall, and weighing 90 kg (Body mass index was 28). He presented pain in the hallux. He presented onychocryptosis in both the medial and lateral canal of the hallux nail and in the medial canal of the second toe nail in the left foot. Although the main reason for the consultation was the onychocryptosis, the patient’s mother stated that for more than a year a prominence had been growing on the dorsal aspect of the head of the first metatarsal of the left foot, and that the patient was limping. The articular examination showed a bilateral increased eversion of the subtalar joint. The first ray of the left foot was dorsiflexed, with palpable dorsal exostosis on the metatarsal head. The first metatarsophalangeal joint showed a lack of extension, with plantarflexion of the proximal phalanx of the hallux. The dynamic examination showed excess of pronation in the right foot during the entire stance phase of gait. The left foot had a total lack of support of the first metatarsal and overload of the hallux. Plantar pressure analysis confirmed the lack of support from the first ray and the excessive pressure on the external part of the foot and on the distal zone of the hallux. On the X-ray exam it was observed that the first ray had become dorsiflexed in a period of a year and a half (Figures 2a and 2b), the time that the patient had been presenting the onychocryptosis. Our hypothesis was that the plantarflexion of the hallux was due to an extreme response to the stimulus from the pain caused by the onychocryptosis, which made the patient adopt an antalgic position of the foot, transferring the weight to the external part of the forefoot, thereby decreasing the load of the first ray. As a result, the proximal phalanx of the hallux became plantarflexed, and this produced the lack of extension of the first metatarsophalangeal joint. The diagnosis was bilateral valgus and flat foot, hallux limitus in the left foot, and onychocryptosis of the first and second toes of the left foot.
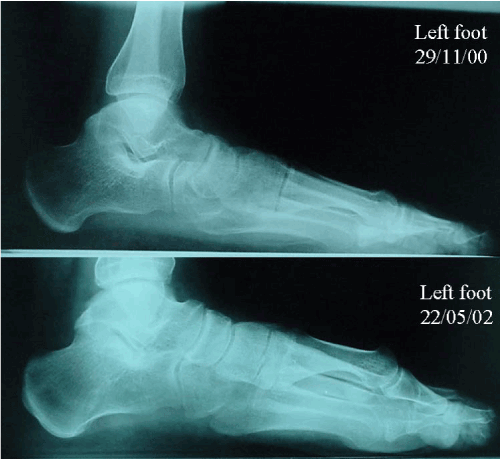
Figure 2. A. The X-ray shows the position of the first ray before the onset of onychocryptosis. B. This image shows the dorsiflexion experimented by the first ray after onset of the onychocryptosis.
The authors’ suspect was that pain caused by the onychocryptosis made the patient adopt an antalgic position of the foot, transferring the body-weight to the external part of the forefoot, thereby decreasing the load capacity of the first ray. Secondarily to this acquired dorsiflexed position of the first ray, the proximal phalanx of the hallux became plantarflexed, producing a hallux limitus [8]. The bibliography consulted stresses that in these patients it is normal that the stimulus-response relationship is altered — particularly the tactile sensations — and if produced in very sensitive areas, such as the distal part of the toe, usually leads to an overstimulation and to avoidance behavior, in this case, of support by the medial edge of the foot.
The treatment proposed combined surgery, kinesiotherapy and orthotics. In order to resolve the process of onychocryptosis definitively, surgical matricectomies of both canals of the first toe and the medial canal of the second were performed with the phenol-alcohol technique. The patient’s convalescence was very good, and cicatrization was complete in a two-week period.
Passive exercises of mobilization of the first metatarsophalangeal joint were prescribed to improve the hallux dorsiflexion. Active exercises to strengthen peroneus longus muscle (the main plantarflexor of the first ray) were prescribed to re-establish the normal load distribution on the foot. The cooperative guidance of his mother was very important of the kinesitherapy.
Custom-designed foot orthoses were applied. Molds were made with plaster bandages under no-load conditions, neutralizing the subtalar joint and extending the hallux to raise the medial arch. Foot orthoses were made of 3-mm thick polypropylene. The one for the right foot had no added modifications. Two orthoses were made for the left foot: One of them had a 5-mm thick semisphere of polyethylene foam fitted to the insole beneath the fifth metatarsal head to provide a lower-intensity stimulus on the external side of the forefoot (similar to the one that the authors’ suspected that had caused the initial problem) that helped load the first metatarsal. This orthotic device was used twice a day for 30 minutes during two months (Figure 3). The constantly-used one was fitted with a valgus wedge in the forefoot to transfer the load to the medial part of the forefoot.
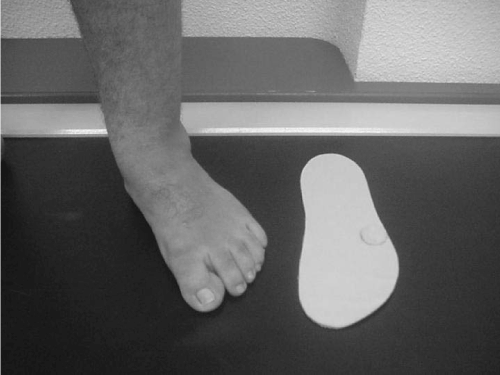
Figure 3. Foot orthosis fitted to the left foot to stimulate the lateral part of the forefoot so that the patient again transfers the weight of the body towards the medial aspect of the forefoot; the arrow is showing the semisphere.
At the four-month follow-up the onychocryptosis was cured, load under the first ray had increased, hallux dorsiflexion had increased 15 degrees, and limping had disappeared.
In this case, a combined therapy was of great importance in obtaining such acceptable results. The authors propose combined treatments and a multi-disciplinary approach to the care of children with rare syndromes as it could avoid short- and long-term problems deriving from an inadequate function of the foot, and improve the quality of life of these patients.
References
- Vicent A, Heitz D, Petit C, Kretz C, Oberlé I, et al. (1991) Abnormal pattern detected in fragile-X patients by pulsed-field gel electrophoresis. Nature 349: 624-6. [Crossref]
2021 Copyright OAT. All rights reserv
- de Vries BB1, Halley DJ, Oostra BA, Niermeijer MF (1998) The fragile X syndrome. J Med Genet 35: 579-589. [CrossRef]
- Ramos FJ (2000) Introducción al Síndrome X Frágil. Boletín de la Federación Española de Asociaciones del Síndrome X Frágil Esp 1: (two pages), 2000.
- De Diego Y (1997) Retraso Mental Hereditario y Síndrome X Frágil. Estudio de afectos y portadores por análisis del ADN (Doctoral Thesis). Sevilla: Universidad de Sevilla.
- Bell MV, Hirst MC, Nakahori Y, MacKinnon RN, Roche A, et al. (1991) Physical mapping across the fragile X: hypermethylation and clinical expression of the fragile X syndrome. Cell 64: 861-866. [CrossRef]
- Willemsen R, Los F, Mohkamsing S, van den Ouweland A, Deelen W, et al. (1997) Rapid antibody test for prenatal diagnosis of fragile X syndrome on amniotic fluid cells: a new appraisal. J Med Genet 34: 250-251. [CrossRef]
- Verkerk AJ, Pieretti M, Sutcliffe JS, Fu YH, Kuhl DP, et al. (1991) Identification of a Gene (FMR-1) Containing a CGG Repeat Coincident with a Breakpoint Cluster Region Exhibiting Length Variation in Fragile X Syndrome. Cell 65: 905-14. [Crossref]
- Lafuente G, González R, Munuera PV (2009) El hallux limitus. In: Munuera PV (Ed.), El primer radio. Biomecánica y ortopodología. Santander: Exa Editores, 193-234.



