Abstract
Basilar artery occlusion accounts for about 1% to 4% of all ischemic strokes and may be related to various causes. Herein we report a case of transient symptomatic basilar artery stenosis secondary to hemoconcentration. A 51-year-old female presented with vertigo, nausea and sudden onset of basilar migraine-like headaches. At admission, Hgb was 17.2 g/dL and Hct 55.2 %. Brain MRI showed a significant flow reduction in the basilar artery and multiple recent ischemic lesions in the brainstem and cerebellum. Serial full blood count checks showed a progressive normalization of hematocrit and hemoglobin levels. At discharge the patient was asymptomatic and a MR-angiography revealed a complete recanalization of the basilar artery. Our report suggests that hemodynamic changes related to hemoconcentration increase the risk of cerebral ischemia and may be reversible if promptly diagnosed and managed.
Keywords
hemoconcentration, cerebral ischemia, headache
Introduction
Basilar artery (BA) occlusion accounts for about 1% to 4% of all ischemic strokes and is generally related to local atherothrombosis or cardioembolism [1]. Hemoconcentration, characterized by increased hematocrit (Hct) and hemoglobin (Hgb) levels, describes the decrease in the ratio between plasma volume and the cellular components of the blood [2]. Herein we report a case of transient symptomatic BA stenosis secondary to hemoconcentration, presenting with sudden onset of basilar migraine-like headaches.
Report of a case
A 51-year-old woman developed sudden onset of headache with migraine features. The headache was bilateral but also intense in the occipital region. It was severe and throbbing and accompanied by nausea and vertigo. If not for the sudden onset of a new headache at a latter age, the case would have been diagnosed as basilar migraine.
However, neurological examination showed a right horizontal nystagmus and a left perceptive hearing loss. She had major depression. Blood pressure was normal during the entire hospital stay. Laboratory investigations at admission found an increase in glycaemia (157 mg/dl; normal 74-106). Hgb was 17.2 g/dL and Hct 55.2%. Common causes of hemoconcentration (i.e myeloproliferative disorders, infections, etc.) were ruled out. Brain MRI at admission showed a significant flow reduction in the BA and multiple recent ischemic lesions in the brainstem and cerebellum (Figure 1). ECG, transthoracic echocardiogram, carotid ultrasound, and genetic analysis of inherited prothrombotic states were normal. The patient was treated with salicylates and had a progressive normalization of Hct and Hgb levels. At discharge she was asymptomatic (headache free) and a MR-angiography revealed a complete recanalization of the BA (Figure 1). Laboratory tests ruled out the presence of dehydration.
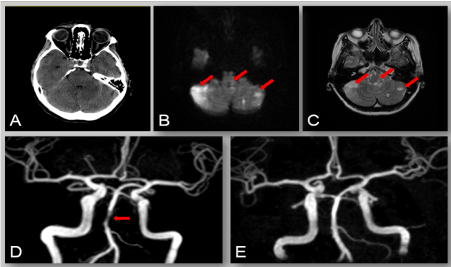
Figure 1: CT (A), MRI (B, C) and MR angiography of the brain (D) at admission. MR-angiography at discharge (E). CT scan was normal (A); axial diffusion-weighted (B) and T2-weighted (C) MR images revealed multiple recent ischemic lesions in the brainstem and both cerebellar hemispheres (arrows). MR angiography at admission (D) showed a significant flow reduction in the middle segment of the BA (arrow). MR-angiography at discharge (E) revealed a complete recanalization of the BA.
Discussion
Hemoconcentration has been documented in depressed subjects and in response to acute mental stress [3,4]. In such conditions, an increased hydrostatic pressure, due to sympathetic nervous system activation, causes movement of fluid into extravascular spaces and consequent concentration of non-diffusible blood components, such as platelets and pro-coagulation factors [3,4]. Furthermore, the increased renal filtration and diuresis following stress conditions can contribute to a prolonged plasma volume loss [4].
Hemoconcentration, due to a polyuria induced by acute secretion of atrial natriuretic peptide, is frequently associated to paroxysmal atrial fibrillation (AF) [5]. Therefore, it can be assumed also that an episode of paroxysmal AF may have caused the ischemic lesions.
The headaches presented by the patient at admission may be explained primarily by a transient intracranial hypertension due to posterior circulation ischemia. Moreover, hemoconcentration alone could explain the headaches, as demonstrated by studies investigating the pathogenesis of headaches during space flights. These studies show that low gravity and consequent hemoconcentration predispose to headaches [6].
Conclusions
Our report suggests that hemodynamic changes related to hemoconcentration increase the risk of cerebral ischemia and may be reversible if promptly diagnosed and managed. The state may present as a new-onset headache and is common in patients with severe psychiatric disease [3,4].
Conflict of interest
The author(s) declare that they have no competing interests.
References
- Mattle HP, Arnold M, Lindsberg PJ, Schonewille WJ, Schroth G (2011) Basilar artery occlusion. Lancet Neurol 10: 1002–1014.
- Allen MT, Patterson SM (1995) Hemoconcentration and stress: a review of physiological mechanisms and relevance for cardiovascular disease risk. Biol Psychol 41: 1-27. [Crossref]
- Wong ML, Dong C, Esposito K, Thakur S, Liu W, et al. (2008) Elevated stress hemoconcentration in major depression is normalized by antidepressant treatment: secondary analysis from a randomized, double-blind clinical trial and relevance to cardiovascular disease risk. PLoS ONE 3: e2350.
- Patterson SM, Krantz DS, Jochum S (1995) Time course and mechanisms of decreased plasma volume during acute psychological stress and postural change in humans. Psychophysiology 32: 538-545. [Crossref]
- Yamada H, Sugiyamam T, Ashida T, Ftjii J (1998) Sustained hemoconcentration in patients with chronic atrial fibrillation, a potential risk for stroke and thromboembolic complications: a retrospective study. Jpn Heart J 39: 715-720. [Crossref]
2021 Copyright OAT. All rights reserv
- Feuerecker M, van Oosterhout WPJ, Feuerecker B, Matzel S, Schelling G, et al. (2016) Headache under simulated microgravity is related to endocrine, fluid distribution, and tight junction changes. Pain 157: 1072-1078. [Crossref]

