Abstract
Introduction
A critical aspect of the physician-modified aortic graft fenestration is represented by the re-sheathing technique which may be complex and not guarantee a correct release of the endograft. In a preliminary experience, we describe an alternative method of opening and closing a physician-modified aortic Gore-Excluder C3 endo-prosthesis. To our knowledge, this method has never been experimented before.
Material and methods
Two protective sheaths with funnel-shaped ends, already present in the graft package, are sectioned to the desired length. The first one is used for the partial and controlled release of the graft body and, after fenestration and distal re-encapsulation, the other is used to complete the re-sheathing of the distal portion of the graft at the hooks level. The endograft forced inside these two sheaths can easily be introduced into a normal Dry-seal and released as usual with a pullback maneuver. No accessory systems / adjunctive techniques or materials are employed.
Over the last 16 months, we have experimented this technique in 6 emergency cases of para-suprarenal abdominal aortic aneurysms, for a total of 11 windows performed (9 renal arteries and 2 AMS). In one case, renal artery perfusion was maintained via a parallel stent-graft (Advanta 8 mm).
Results
The average graft preparation-time was 46' (min. 30'- max. 70'). In all cases, graft-release was performed correctly with normal perfusion of fenestrations. The CT angiography documented the absence of graft-related complications in all cases at 6-month follow-up (2-12).
Conclusions
This original and simple technique has proven to be effective and seems to offer real advantages in terms of simplicity and execution time compared to others previously described. The possibility of using the original introducer according to the usual technique and the typical re-positioning of the Gore Excluder C3 endoprosthesis are preserved; this specific characteristic of the Endograft significantly facilitates the re-sheathing maneuver. A more extensive experience is mandatory to assess the reproducibility of these results.
Keywords
endovascular aortic repair, Gore-Excluder C3 endoprostheses, juxta-renal abdominal aortic aneurysm, physician-modified aortic endograft fenestration, surgeon-modified stent-graft, technique of partial deployment and re-sheathing of physician-modified aortic endograft
Introduction
Since 2012 the Cook Z stent aortic graft (Cook Medical Inc, Bloomington, Ind), has been the only device available for physician-modified fenestration because its constraining wire allowed physicians to re-sheath the device after modifications.
The Gore Excluder C3 endograft was more recently proposed as an alternative option for physician-modified endovascular aneurysm repair for its ability to partially deploy and subsequently re-sheath the graft (W. L. Gore & Associates, Inc, Flagstaff, Ariz). This technique was first proposed in 2012 by Paludetto and accurately described step by step by Coulter et al. in 2014 [1].
While it has been reported that the immediate results of physician-modified endografts for prevention of aneurysm-related deaths are comparable to manufactured devices, the long-term outcome of this technique remains undetermined [2,3].
Actually, one critical aspect of this procedure is represented by the re-sheathing technique and specifically, by the possibility to partially deploy and successively properly reconstitute the graft [1,4,5].
In fact, this manoeuvre can be complex, time consuming and, above all, not guarantee the correct endo-graft release.
In a preliminary experience, we describe an original technique of partial release and re-sheathing of a physician-modified aortic Gore-Excluder C3 endoprostheses.
To our knowledge, this method of physician-modified aortic endograft fenestration has never been experienced until today.
Material and methods
Step by step Technique description
endograft modification using Gore Excluder C3 endograft was performed under strict sterile conditions on a back table just before graft implantation. Two protective sheaths with funnel-shaped ends already present in the Gore Excluder C3 endoprosthesis package, are employed. These are cut to a desired length (leaving the funnel-shaped side intact), to obtain two endograft re-sheathing systems (ERS) (Figure 1A). The first is used for the partial and controlled release of the endoprosthetic body and its distal re-encapsulation and is preloaded on the delivery system with the funnel-shaped side facing towards the tip of the graft. The second one is used to complete graft re-sheathing in its proximal area at the hooks level. The main body of the endograft is partially unsheathed in the main trunk portion; the contralateral gate is kept constrained by the ERS (Figures 1B and C). Fenestrations are made by sharply incising the ePTFE, followed by a circumferential reinforcement suture with a cut gooseneck wire. The locations for the fenestrations were pre-marked on the body of the stent graft using the measurements obtained from the centre-line analysis on CTA. The deployed portion of the Endograft is partially re-sheathed into the distal ERS by pushing it forward. The nitinol barb stents are left free (Figures 2A-D). The Endograft re-sheathing is completed by introducing the second ERS from the tip until the funnel-shaped ends touch each other. This maneuver is significantly facilitated by the re-positioning deployment system of the Gore Excluder C 3 Endograft. Both ERS are moved distally. The prosthesis forced inside these sheaths can be introduced as usual into a normal Dryseal (Figures 3 A-D) and subsequently released with a pull-back maneuver. No accessory other systems / adjunctive techniques / materials are employed for the re-constriction of the graft.

Figure 1. A: two protective sheaths with funnel-shaped ends already present in the Gore Excluder C3 endoprosthesis package, are cut to a desired length leaving the funnel-shaped side intact to obtain two endograft re-sheathing systems (ERS). The on table demonstration shows how the ERSs are properly positioned with the funnel-shaped ends that touch each other (circle); B,C: The first ERS is used for the partial and controlled unsheathing of the main trunk portion of the graft (large arrow)
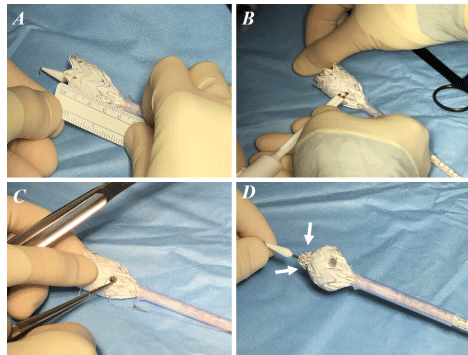
Figure 2. A,B,C: The locations for the fenestrations were pre-marked on the body of the stent graft using the measurements obtained from the center-line analysis on CTA. Fenestrations are made by sharply incising the ePTFE, followed by a circumferential reinforcement suture with a cut gooseneck wire; D: The deployed portion of the Endograft is partially re-sheathed into the distal protective ERS by pushing it forward. The nitinol barb stents are closed again by the Gore Excluder C3 repositionability deployment system and left free (small arrows).
Preliminary clinical experience
Over the last 16 months we have adopted this technique in 6 cases of para or suprarenal aortic aneurysms, all performed in emergency conditions for impending rupture. In all cases the risks and benefits of different treatment options were discussed at length with the patients who fully understood that fenestrated endovascular aneurysm repair with physician-modified stent grafts was off-label use and consented to proceed with the procedure. Also in all considered cases, A CT-Angiogram evidenced a juxta-renal AAA with “no neck zone” under the lower renal artery and the suprarenal aorta suitable for endograft positioning. Axial images were used to identify the position of the renal arteries / mesenteric vessels by use of clock-face orientation. A total of 11 windows were performed (9 for the renal arteries and 2 for the AMS). In one case, the perfusion of one renal artery was maintained via a parallel stent (Advanta 8 mm). A covered stent-graft (Begraft) was deployed in all cases into the fenestrations and flared proximally with an 8-mm angioplasty balloon.
Results
The average preparation time of the prosthesis was 46' (min. 30' – max. 70'). In all cases, graft release was performed correctly and the conserved possibility of re-positioning the endoprosthesis allowed the ideal perfusion of all windows and target vessels. In all cases, the completion angiogram demonstrated the patency of all target vessels in absence of Type one endoleak and the CT angiography control documented the absence of graft-related complications at an average of 6-month follow-up (min. 2 – max. 12).
Discussion
Back table modification of endografts began as early as 2006 [6], while the term physician-modified endografts was first proposed by Starnes in 2012 in order to define physician modifications of manufactured abdominal and thoracic stent grafts with the creation of fenestrations and directional branches [2,7].
This technique primarily experienced with the Zenith platform (Cook Medical) was successively extended with modifications to further commercially available endovascular grafts [1,3,8]. More recently, it was also proposed for inner branched endograft modification in cases of emergency thoraco-abdominal aneurysm repair [9,10].
Recent publications have shown that technical and clinical results of physician-modified endografts are similar to patient-specific devices [11].
Oderich et al in a recent retrospective study (between 2007 and 2016) on 316 consecutive patients who underwent fenestrated-branched endovascular aortic repair with a physician-modified endograft or a custom-made device, documented a higher mortality and morbidity rate for the physician-modified endograft series at 3-year follow-up. These results are most probably related to the specific clinical characteristics of this group of patients. Conversely, patient survival, as well as freedom from reintervention, primary and secondary target vessel patency, and the absence of renal function deterioration were similar in the two considered series. Furthermore, the same study documented that both techniques were equally effective for late prevention of aneurysm-related deaths [2].
However, long-term outcome of this technique remains uncertain and, similarly, the long-term effects of graft modification on endograft structure and strength are unknown; consequently today this technique is still considered “off-label” and should be reserved to patients not candidates for a custom-made device for the lack of preparation time as in cases of emergency indication of endovascular aneurysm repair (due to rapid sac expansion, impending or contained aneurysm rupture) [2,12].
In our series, all included patients presented a juxta-renal abdominal aortic aneurysm and all were operated in emergency conditions for impending aneurysm rupture.
One of the main specific disadvantages of the physician-modified fenestration technique is represented by the fact that graft modification may be complex and “time consuming”.
Specifically, it has been reported that the average modification time for the most frequently experienced Zenith Alpha thoracic stent-graft ranged from one to two hours; also, this drawback significantly limits the larger application of this technique [10].
In these cases, the extensive preparation time is also related to the need to employ accessory systems or adjunctive maneuvers or materials to partially relay and re-sheath the endograft correctly.
This original technique of partial deployment and re-sheathing of a physician-modified aortic Gore-Excluder C3 endoprosthesis has significantly reduced the average graft preparation time which resulted between 30’ to 70’ (mean 46’) in this preliminary experience.
We believe that the relatively short preparation time is mainly related to its simplicity based on the accurate and careful use as ERS of two protective sheaths with funnel-shaped ends, already present in the Gore Excluder C3 endoprosthesis package, modified and sectioned to the desired length.
Ultimately, in our opinion, it should be stressed that the Endograft re-sheathing in the second ERS from the tip of the endograft (until the funnel-shaped ends touch each other), is significantly facilitated by the unique characteristic of the re-positioning system of the Gore Excluder C3 Endograft (Figures 2 and 3)
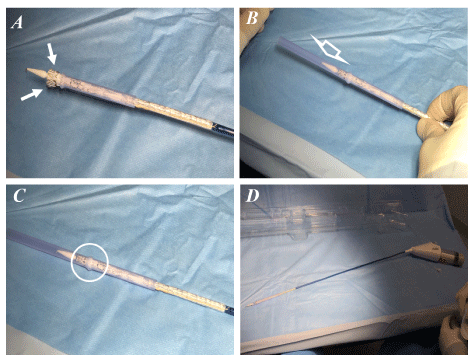
Figure 3. A,B,C: A, The Endograft re-sheathing in its proximal area at the hooks level (small arrows) is completed by introducing the second protective ERS (large arrow) from the tip until the funnel-shaped ends touch each other (circle); D, both protective ERS moved distally. The prosthesis forced inside these sheaths can be introduced as usual into a normal Dryseal and subsequently released with a pull-back maneuver. ERS, endograft re-sheathing systems.
In conclusion, we believe that this original technique might represent the “Columbus’ egg” of physician-modified fenestration practice.
This simple and unique system of partial delivery and re-sheathing of a physician-modified Gore C3 endograft (using only the contents of the endograft package), has proven effective in our preliminary experience and seems to offer significant advantages in terms of simplicity and speed of execution compared to others described previously.
The possibility of using the original introducer according to the usual technique and the typical re-positioning of the Gore Excluder C3 endoprosthesis are preserved. This specific characteristic of the Endograft significantly facilitates the re-sheathing maneuver.
Only a broader experience and long-term follow-up may confirm the reproducibility of these results.
Acknowledgement
- The authors wish to thank Ms. Elena Harwood for the linguistic revision of this paper.
- The authors declare that they have no competing interests.
- Informed consent was taken by the patient / No funding received
References
- Coulter AH, Tan TW, Zhang WW (2014) Physician modification of Gore C3 excluder endograft for treatment of abdominal aortic aneurysms anatomically unsuitable for conventional endovascular repair. J Vasc Surg 59: 1739-1743. [Crossref]
- Oderich GS, Ribeiro MS, Sandri GA, Tenorio ER, Hofer JM, et al. (2019) Evolution from physician-modified to company-manufactured fenestrated-branched endografts to treat pararenal and thoracoabdominal aortic aneurysms. J Vasc Surg 70: 31-42. [Crossref]
- Georgiadis GS, van Herwaarden JA, Antoniou GA, Constantijn EV, Hazenberg B, et al. (2016) Systematic review of off-the-shelf or physician-modified fenestrated and branched endografts. J Endovasc Ther 1: 98-109. [Crossref]
- Oderich GS (2010) Technique of adding a diameter-reducing wire to the modified TX2 fenestrated stent graft. Vascular 6: 350-355. [Crossref]
- Oderich GS, Mendes BC, Correa MP (2014) Preloaded guidewires to facilitate endovascular repair of thoracoabdominal aortic aneurysm using a physician-modified branched stent-graft. J Vasc Surg 4: 1168-1173. [Crossref]
- Uflacker R, Robison JD, Schonholz C, Ivancev K (2006) Clinical experience with a customized fenestrated endograft for juxtarenal abdominal aortic aneurysm repair. J Vasc Interv Radiol 12: 1935-1942. [Crossref]
- Starnes BW (2012) Physician-modified endovascular grafts for the treatment of elective, symptomatic, or ruptured juxtarenal aortic aneurysms. J Vasc Surg 3: 601-607. [Crossref]
- Tsilimparis N, Banafsche R, Stana J (2020) Physician-Modified Endografts: Surmounting Anatomical Challenges with Innovative Techniques to Optimize Treatment. J Endovasc Ther 1: 130-131. [Crossref]
- Shibata T, Kawaharada N, Yasuhara K, Naraoka S (2022) Physician-modified stent graft with inner branches for treating ruptured thoracoabdominal aortic aneurysm. Eur J Cardio-Thoracic Surg 4: 952-954. [Crossref]
- D’Oria M, Mirza AK, Tenorio ER, Kärkkäinen JM, DeMartino RR, et al. (2020) Physician-Modified Endograft With Double Inner Branches for Repair of Supraceliac Para-Anastomotic pseudoaneurysm. J Endovasc Ther 1: 124 -129. [Crossref]
- Fernandez CC, Sobel JD, Gasper WJ, Vartanian SM, Reilly RM, et al. (2016) Standard off-the shelf versus custom-made multibranched thoracoabdominal aortic stent grafts. J Vasc Surg 63: 1208-1215. [Crossref]
- Singh A, Mafeld S, Williams R, Mc Caslin J (2018) Physician-Modified Fenestrated Endografts for Managing the Ruptured or Symptomatic Aortic Aneurysm: Technique Overview and Clinical Outcomes. Vasc Endovasc Surg 52: 607-612. [Crossref]



